Tesamorelin Quick Start
Tesamorelin is the only FDA-approved GHRH (growth hormone-releasing hormone) peptide. It signals your pituitary gland to release more of your own growth hormone in its natural rhythm. It is sold as Egrifta SV and Egrifta WR for HIV-related belly fat (lipodystrophy). It is also sold as research-grade vials for off-label study.
Most protocols use 2 mg once a day under the skin of the abdomen. Egrifta SV uses 1.4 mg daily and Egrifta WR uses 1.28 mg daily. Research vials usually come in 2 mg, 5 mg, or 10 mg sizes and are mixed with bacteriostatic water before use.
Route
Subcutaneous injection in the abdomen only. Rotate sites daily.
Schedule
Once daily. No ramp-up. Full dose from day one.
Measure
Use a U-100 insulin syringe. Volume depends on vial concentration after reconstitution.
Supplies
Peptide vials, bacteriostatic water, syringes, and alcohol swabs.
Research status
FDA-approved for HIV lipodystrophy. All other use is off-label.
Disclaimer
This page is an educational research reference and is not medical advice. Tesamorelin is FDA-approved only for HIV-associated lipodystrophy. Use for any other purpose is off-label and should involve a licensed clinician.
This page covers Tesamorelin dose steps, timing, vial math, and storage. Want the full research background? Read the Tesamorelin guide. It covers how it works, study results, side effects, and legal status.
Tesamorelin Dosing Protocol & Schedule
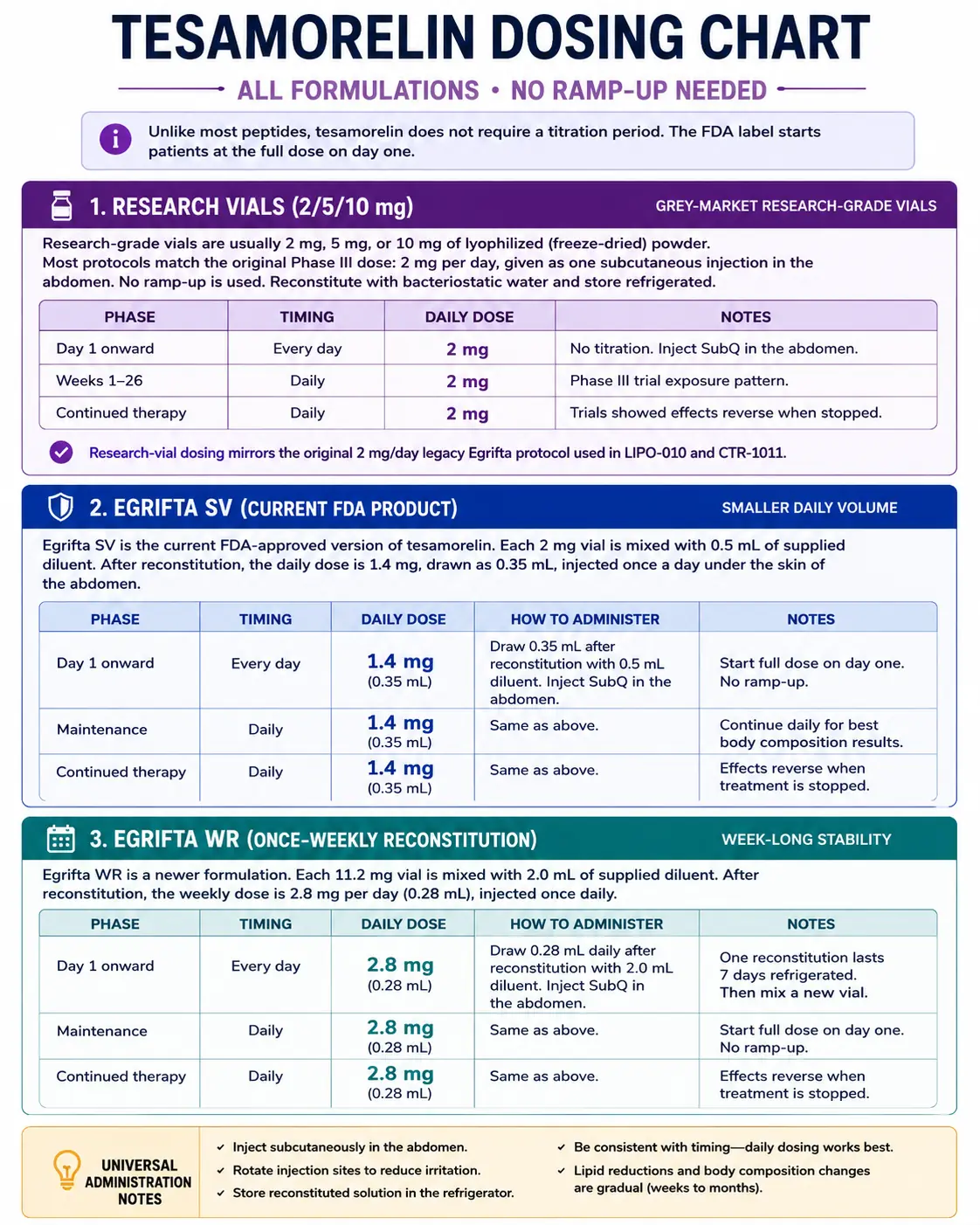
Unlike most peptides, tesamorelin does not need a slow ramp-up. The FDA label starts patients at the full dose on day one. The table below shows dosing for all three formulations: Egrifta SV (the current main product), Egrifta WR (a newer once-weekly-reconstitution version), and legacy Egrifta (the original 2 mg/day product used in trials). Research vials from grey-market sources are usually dosed like legacy Egrifta — 2 mg once a day.
Tesamorelin Dosing by Formulation
Choose the formulation you are researching to see the matching daily dose and prep notes.
Grey-market research-grade vials.
Research-grade vials are usually 2 mg, 5 mg, or 10 mg of lyophilized (freeze-dried) powder. Most protocols match the original Phase III dose: 2 mg per day, given as one subcutaneous injection in the abdomen. No ramp-up is used. Reconstitute with bacteriostatic water and store refrigerated.
Research Vial Dosing — Phase III Equivalent
Phase
Day 1 onward
Timing
Every day
Daily Dose
2 mg
Notes
No titration. Inject SubQ in the abdomen.
Phase
Weeks 1-26
Timing
Daily
Daily Dose
2 mg
Notes
Phase III trial exposure pattern.
Phase
Continued therapy
Timing
Daily
Daily Dose
2 mg
Notes
Trials showed effects reverse when stopped.
| Phase | Timing | Daily Dose | Notes |
|---|---|---|---|
| Day 1 onward | Every day | 2 mg | No titration. Inject SubQ in the abdomen. |
| Weeks 1-26 | Daily | 2 mg | Phase III trial exposure pattern. |
| Continued therapy | Daily | 2 mg | Trials showed effects reverse when stopped. |
Research-vial dosing mirrors the original 2 mg/day legacy Egrifta protocol used in LIPO-010 and CTR-1011.
Current FDA product with smaller daily volume.
Egrifta SV is the current FDA-approved version of tesamorelin. Each 2 mg vial is mixed with 0.5 mL of supplied diluent. After reconstitution, the daily dose is 1.4 mg, drawn as 0.35 mL, injected once a day under the skin of the abdomen.
Egrifta SV — Label Dosing
Phase
Day 1 onward
Timing
Every day
Daily Dose
1.4 mg
Notes
Draw 0.35 mL after reconstitution with 0.5 mL diluent.
Phase
Maintenance
Timing
Daily
Daily Dose
1.4 mg
Notes
Use immediately after mixing (per label).
| Phase | Timing | Daily Dose | Notes |
|---|---|---|---|
| Day 1 onward | Every day | 1.4 mg | Draw 0.35 mL after reconstitution with 0.5 mL diluent. |
| Maintenance | Daily | 1.4 mg | Use immediately after mixing (per label). |
Egrifta SV is single-use. The label says to inject right after reconstitution.
Newer weekly-reconstitution format.
Egrifta WR is the newest FDA-approved format. The 11.6 mg vial is mixed once a week with 1.45 mL of bacteriostatic water. The daily dose is 1.28 mg, drawn as 0.16 mL, injected once a day under the skin of the abdomen.
Egrifta WR — Label Dosing
Phase
Day 1 onward
Timing
Every day
Daily Dose
1.28 mg
Notes
Draw 0.16 mL after weekly reconstitution.
Phase
Maintenance
Timing
Daily
Daily Dose
1.28 mg
Notes
Store reconstituted vial at 2-8C for up to 28 days.
| Phase | Timing | Daily Dose | Notes |
|---|---|---|---|
| Day 1 onward | Every day | 1.28 mg | Draw 0.16 mL after weekly reconstitution. |
| Maintenance | Daily | 1.28 mg | Store reconstituted vial at 2-8C for up to 28 days. |
Egrifta WR cuts injection prep down to once per week instead of daily.
Cycle Guidelines
Tesamorelin Cycle Guidelines
Approach
Standard
Duration
26 weeks
Review Point
Week 26 imaging
Best For
Phase III trial exposure pattern
Approach
Extended
Duration
52 weeks
Review Point
Week 52 imaging
Best For
Pooled Phase III extension data
Approach
Long-term (NAFLD context)
Duration
12 months
Review Point
12-month imaging
Best For
Liver-fat study exposure pattern
| Approach | Duration | Review Point | Best For |
|---|---|---|---|
| Standard | 26 weeks | Week 26 imaging | Phase III trial exposure pattern |
| Extended | 52 weeks | Week 52 imaging | Pooled Phase III extension data |
| Long-term (NAFLD context) | 12 months | 12-month imaging | Liver-fat study exposure pattern |
Trial data showed visceral fat returns toward baseline after stopping. Sustained therapy is generally needed to keep results.
Important Dosing Notes
No titration required. The label and Phase III trials started patients at the full dose on day one.
Injection site. Only the abdomen is on the label. Rotate spots and avoid scarred or bruised tissue.
Timing. Tesamorelin can be given any time of day. Trials were not bedtime-only. Some users pick morning; others pick bedtime.
Missed dose. Skip the missed dose. Resume the next scheduled day. Do not double up.
Formulation note. Grey-market research vials use bacteriostatic water and are stored refrigerated. Egrifta SV uses its own supplied diluent and is used right after mixing.
Tesamorelin Reconstitution Guide
Reconstitution means mixing the dry powder in the vial with bacteriostatic water (BAC water) to make a liquid you can draw into a syringe. The amount of water you add changes the concentration — less water means a stronger mix and smaller injections, more water means a weaker mix and easier-to-measure injections.
The table below shows mixing options for the most common research vial sizes. Find your vial size on the left, pick a BAC water volume, then read across to your target dose.
Tesamorelin Reconstitution — Vial Size, BAC Water, and Dose Volumes
Vial Size
2 mg
BAC Water
0.5 mL
Concentration
4,000 mcg/mL
1 mg Dose
0.25 mL (25 units)
1.4 mg Dose
0.35 mL (35 units)
2 mg Dose
0.50 mL (50 units, full vial)
Vial Size
2 mg
BAC Water
1.0 mL
Concentration
2,000 mcg/mL
1 mg Dose
0.50 mL (50 units)
1.4 mg Dose
0.70 mL (70 units)
2 mg Dose
1.0 mL (100 units, full vial)
Vial Size
5 mg
BAC Water
1.0 mL
Concentration
5,000 mcg/mL
1 mg Dose
0.20 mL (20 units)
1.4 mg Dose
0.28 mL (28 units)
2 mg Dose
0.40 mL (40 units)
Vial Size
5 mg
BAC Water
2.5 mL
Concentration
2,000 mcg/mL
1 mg Dose
0.50 mL (50 units)
1.4 mg Dose
0.70 mL (70 units)
2 mg Dose
1.0 mL (100 units)
Vial Size
10 mg
BAC Water
2.0 mL
Concentration
5,000 mcg/mL
1 mg Dose
0.20 mL (20 units)
1.4 mg Dose
0.28 mL (28 units)
2 mg Dose
0.40 mL (40 units)
Vial Size
10 mg
BAC Water
5.0 mL
Concentration
2,000 mcg/mL
1 mg Dose
0.50 mL (50 units)
1.4 mg Dose
0.70 mL (70 units)
2 mg Dose
1.0 mL (100 units)
| Vial Size | BAC Water | Concentration | 1 mg Dose | 1.4 mg Dose | 2 mg Dose |
|---|---|---|---|---|---|
| 2 mg | 0.5 mL | 4,000 mcg/mL | 0.25 mL (25 units) | 0.35 mL (35 units) | 0.50 mL (50 units, full vial) |
| 2 mg | 1.0 mL | 2,000 mcg/mL | 0.50 mL (50 units) | 0.70 mL (70 units) | 1.0 mL (100 units, full vial) |
| 5 mg | 1.0 mL | 5,000 mcg/mL | 0.20 mL (20 units) | 0.28 mL (28 units) | 0.40 mL (40 units) |
| 5 mg | 2.5 mL | 2,000 mcg/mL | 0.50 mL (50 units) | 0.70 mL (70 units) | 1.0 mL (100 units) |
| 10 mg | 2.0 mL | 5,000 mcg/mL | 0.20 mL (20 units) | 0.28 mL (28 units) | 0.40 mL (40 units) |
| 10 mg | 5.0 mL | 2,000 mcg/mL | 0.50 mL (50 units) | 0.70 mL (70 units) | 1.0 mL (100 units) |
U-100 insulin syringes have 100 units = 1.0 mL.
Step-by-Step Reconstitution
- 01
Clean the workspace
Swab the vial stopper with alcohol. Let it air dry for about 10 seconds.
- 02
Draw the BAC water
Use a fresh sterile syringe. Pull up the volume of bacteriostatic water from the table above.
- 03
Add water against the wall
Push the needle through the stopper. Inject the water slowly down the side of the vial, not directly onto the powder.
- 04
Let it flow
Allow the water to wash gently over the powder. No pressure spray.
- 05
Roll, do not shake
Gently roll the vial between your hands for 30 to 60 seconds until fully dissolved.
- 06
Inspect the solution
Use only if the liquid is clear and colorless. Discard if cloudy or if you see particles.
- 07
Label and refrigerate
Mark the vial with concentration and reconstitution date. Store at 2-8C. (Egrifta SV is single-use — inject right after mixing per the label.)
Need custom math?
Use the free Peptide Reconstitution Calculator to get exact syringe units for any vial size and BAC water volume.
Tesamorelin Dosage Chart
This tesamorelin dosage chart summarizes the no-ramp-up daily dosing approach across research vials, Egrifta SV, and Egrifta WR, with dose and administration notes by formulation.
How Tesamorelin Works
Tesamorelin tells your pituitary gland (the small gland in your brain that controls growth hormone) to release more growth hormone (GH) in its natural rhythm. That extra GH then drives the body to break down deep belly fat and liver fat more than other fat stores.
GHRH Receptor Binding
Your pituitary has docking points called GHRH receptors. Tesamorelin locks onto these receptors and turns on the same signaling pathways (cAMP and PKA) your body uses naturally. The result is a normal-shaped pulse of GH release, not a constant flood.
IGF-1 Increase
Once GH enters the bloodstream, the liver makes more IGF-1 (insulin-like growth factor 1). IGF-1 carries out many of GH's downstream effects on fat and tissue. Trials show IGF-1 rises while other pituitary hormones stay in their normal ranges.
Fat Breakdown
The GH and IGF-1 rise drives fat breakdown in visceral fat (the deep fat around your organs) more than in fat under the skin. It also reduces fat buildup in the liver. Phase III trials and NAFLD studies confirmed these effects, which is what earned tesamorelin its FDA approval.
Feedback Stays Intact
Unlike injecting growth hormone directly (which can shut off your body's own GH production), tesamorelin keeps your normal feedback loops. Your brain still controls the system. Tesamorelin only amplifies the signal your body already sends.
Tesamorelin Supplies Needed
Use the dosing schedule above to plan supplies. Math below assumes the research-vial protocol: 2 mg per day from a 10 mg vial reconstituted with 2.0 mL bacteriostatic water (5,000 mcg/mL concentration, 0.40 mL / 40 units per dose).
Recommended Supply
PEPPAL applies to eligible supplier checkout links when supported by the supplier.

Tesamorelin Vials

SiPhox Health At-Home Blood Test
Injection Supplies
Disclosure: supply links may earn PDP a commission at no cost to you.
Peptide Vials
Math assumes 10 mg vials at 2 mg/day. One 10 mg vial covers about 5 doses.
| Cycle length | Planning note |
|---|---|
4 weeks 6 vials (10 mg) | 28 daily doses needed. Allow ~5 doses per vial. |
8 weeks 12 vials (10 mg) | 56 daily doses needed. |
12 weeks 17 vials (10 mg) | 84 daily doses needed. |
26 weeks 37 vials (10 mg) | 182 daily doses needed. Matches Phase III exposure. |
4 weeks
6 vials (10 mg)
28 daily doses needed. Allow ~5 doses per vial.
8 weeks
12 vials (10 mg)
56 daily doses needed.
12 weeks
17 vials (10 mg)
84 daily doses needed.
26 weeks
37 vials (10 mg)
182 daily doses needed. Matches Phase III exposure.
Insulin Syringes (U-100)
Use 0.5 mL / 50-unit syringes. One syringe per daily injection.
| Cycle length | Planning note |
|---|---|
4 weeks 28 syringes | 1 syringe per injection. |
8 weeks 56 syringes | 1 syringe per injection. |
12 weeks 84 syringes | 1 syringe per injection. |
26 weeks 182 syringes | 1 syringe per injection. |
4 weeks
28 syringes
1 syringe per injection.
8 weeks
56 syringes
1 syringe per injection.
12 weeks
84 syringes
1 syringe per injection.
26 weeks
182 syringes
1 syringe per injection.
Bacteriostatic Water
Use 2.0 mL per 10 mg vial. Each 10 mL bottle covers about 5 vials.
| Cycle length | Planning note |
|---|---|
4 weeks 2 x 10 mL bottles | 6 vials use 12 mL total. Round up. |
8 weeks 3 x 10 mL bottles | 12 vials use 24 mL total. |
12 weeks 4 x 10 mL bottles | 17 vials use 34 mL total. |
26 weeks 8 x 10 mL bottles | 37 vials use 74 mL total. |
4 weeks
2 x 10 mL bottles
6 vials use 12 mL total. Round up.
8 weeks
3 x 10 mL bottles
12 vials use 24 mL total.
12 weeks
4 x 10 mL bottles
17 vials use 34 mL total.
26 weeks
8 x 10 mL bottles
37 vials use 74 mL total.
Round up to allow for priming losses, dropped syringes, damaged swabs, and any protocol adjustments.
Who Tesamorelin Is For and Who Should Avoid It
Tesamorelin (Egrifta) is FDA-approved for adults with HIV-associated lipodystrophy — meaning people on HIV medication who have built up excess belly fat. Use for any other reason is off-label and should involve a licensed clinician.
Should Not Use Tesamorelin
The FDA label lists several groups who should not use tesamorelin. These include people with active cancer (any type), people who are pregnant or planning pregnancy, anyone with a known allergy to tesamorelin or mannitol (an inactive ingredient), and people with major problems of the hypothalamus or pituitary gland (the brain area that controls hormone release).
Use With Caution
Some groups should be reviewed carefully with a clinician before starting. These include people with type 2 diabetes or higher blood sugar, people with a history of cancer, and people with eye conditions like diabetic retinopathy. The label recommends checking blood sugar before starting and during use because GH can affect glucose levels.
Pregnancy and Breastfeeding
Tesamorelin is not safe in pregnancy. It can also cross into breast milk. The label recommends stopping tesamorelin if pregnancy is suspected.
Tesamorelin Side Effects & Safety
Tesamorelin has one of the largest safety databases of any peptide. Phase III trials covered more than 800 participants and tracked side effects through 26 to 52 weeks of daily injections.
Most Common Side Effects
Injection-site reactions are the most common. These include redness, itching, pain, swelling, and rash. In CTR-1011, injection-site issues happened in 50.7% of tesamorelin users versus 21.4% on placebo. In LIPO-010, the numbers were 30.0% versus 24.1%.
Fluid retention, joint pain, and muscle aches can also happen because of GH-related fluid shifts. Swelling occurred in about 9.9% of tesamorelin users versus 5.8% on placebo. Muscle aches occurred in 3.7-7.7% versus 1.6-2.2% on placebo. Most cases were mild to moderate.
Blood Sugar Changes
Trials showed more tesamorelin users had elevated HbA1c (a 3-month average blood sugar marker) compared to placebo. The FDA label recommends checking blood sugar before starting and at intervals during use.
Antibody Development
About half of trial participants developed anti-tesamorelin IgG antibodies after 26 to 52 weeks. The good news: visceral fat reduction and IGF-1 effects were similar in people who developed antibodies and those who did not.
IGF-1 Levels
IGF-1 is expected to rise on tesamorelin. Persistent very high levels (for example, sustained above 3 SDS in clinical context) may need clinician review, dose adjustment, or stopping.
Should Not Use Tesamorelin
Active cancer, pregnancy, known allergy (including to mannitol), and major problems of the hypothalamus or pituitary gland are listed contraindications. See the PepPal Peptide Side Effects Guide for a broader cross-class safety review.
Tesamorelin Timeline & What to Monitor
Tesamorelin is a slow-acting peptide. Trial outcomes were measured in weeks and months, not days. Below is a rough timeline based on Phase III and NAFLD trial data, plus the markers that make sense to track with a clinician.
Tesamorelin Timeline — What Trials Measured and When
Time Point
Week 2
What Trials Saw
IGF-1 levels rise; overnight GH output goes up (Grinspoon 2011 healthy-male data).
Time Point
Week 13
What Trials Saw
Early visceral fat decrease begins (interim trial readouts).
Time Point
Week 26
What Trials Saw
Phase III primary endpoint: VAT reduction of 11.7 to 15.2% versus placebo.
Time Point
Week 52
What Trials Saw
Pooled extension data showed maintained VAT effects with continued therapy.
Time Point
12 months
What Trials Saw
Lancet HIV NAFLD trial: liver-fat reduction of about 32% versus placebo, with lower fibrosis progression.
Time Point
After stopping
What Trials Saw
VAT and other effects trended back toward baseline.
| Time Point | What Trials Saw |
|---|---|
| Week 2 | IGF-1 levels rise; overnight GH output goes up (Grinspoon 2011 healthy-male data). |
| Week 13 | Early visceral fat decrease begins (interim trial readouts). |
| Week 26 | Phase III primary endpoint: VAT reduction of 11.7 to 15.2% versus placebo. |
| Week 52 | Pooled extension data showed maintained VAT effects with continued therapy. |
| 12 months | Lancet HIV NAFLD trial: liver-fat reduction of about 32% versus placebo, with lower fibrosis progression. |
| After stopping | VAT and other effects trended back toward baseline. |
Trial data; not a promise of personal results.
What to Monitor With a Clinician
Reasonable markers to track include fasting blood sugar, HbA1c, IGF-1, liver enzymes (ALT, AST), and lipid panel. Body composition can be tracked with waist circumference or DEXA imaging if available. Stop and review with a clinician if blood sugar climbs significantly, if IGF-1 rises far above normal, or if side effects do not settle.
Tesamorelin Clinical Evidence Context
Tesamorelin has one of the strongest clinical programs in peptide therapeutics. Two large Phase III trials, a Lancet HIV NAFLD study, and mechanistic transcriptomic and proteomic follow-up papers anchor the evidence base.
LIPO-010 (Phase III, n=412)
Falutz et al. HIV lipodystrophy patients showed about 15.2% visceral fat reduction at 26 weeks versus placebo, plus favorable lipid and trunk-fat changes.
CTR-1011 (Phase III, n=404)
Falutz et al. Replication trial showed about 11.7% VAT reduction at 26 weeks, with increased lean mass and waist changes.
Pooled Phase III analysis (n=816)
Consistent VAT reductions across both trials with maintenance during continued therapy and regression toward baseline after stopping.
Stanley et al. 2019 (Lancet HIV, n=61)
Phase II investigator-initiated trial in HIV-NAFLD: about 32% liver-fat reduction and lower fibrosis-progression rate versus placebo at 12 months.
Stanley et al. 2014 (JAMA, n=54)
Phase II trial in HIV with abdominal fat: significant VAT reduction over 6 months with metabolic and hepatic-signal analyses.
Fourman et al. 2020 (JCI Insight)
Mechanistic follow-up: tesamorelin shifted liver transcriptomic signatures toward oxidative phosphorylation and reduced inflammatory signaling.
Fourman et al. 2021 (Scientific Reports)
Proteomic profiles identified response pathways tied to inflammation and tissue-remodeling modulation in HIV-NAFLD.
Grinspoon et al. 2011 (Phase I, n=13)
Healthy men: increased overnight GH output and pulse-area effects with reversal after withdrawal.
Falutz et al. 2024 (AIDS)
Efficacy and safety review of tesamorelin in people with HIV on integrase inhibitors — confirms continued relevance in modern HIV care.
Limitations
Direct trial evidence is in HIV-associated populations. Off-label use in non-HIV populations relies on extrapolation, not large RCTs.
Tesamorelin Storage & Handling
Tesamorelin Storage
State
Lyophilized Egrifta SV
Storage
77F (25C) controlled room temp
Duration
Until expiration. 59-86F (15-30C) excursions allowed.
State
Lyophilized research vials
Storage
-4F (-20C) freezer
Duration
Long-term storage (years).
State
Lyophilized research vials
Storage
35.6-46.4F (2-8C) refrigerator
Duration
Several months.
State
Reconstituted Egrifta SV
Storage
Use immediately
Duration
Discard if not used right away (label).
State
Reconstituted Egrifta WR
Storage
35.6-46.4F (2-8C) refrigerator
Duration
Up to 28 days.
State
Reconstituted research vials
Storage
35.6-46.4F (2-8C) refrigerator
Duration
Up to 28 days.
| State | Storage | Duration |
|---|---|---|
| Lyophilized Egrifta SV | 77F (25C) controlled room temp | Until expiration. 59-86F (15-30C) excursions allowed. |
| Lyophilized research vials | -4F (-20C) freezer | Long-term storage (years). |
| Lyophilized research vials | 35.6-46.4F (2-8C) refrigerator | Several months. |
| Reconstituted Egrifta SV | Use immediately | Discard if not used right away (label). |
| Reconstituted Egrifta WR | 35.6-46.4F (2-8C) refrigerator | Up to 28 days. |
| Reconstituted research vials | 35.6-46.4F (2-8C) refrigerator | Up to 28 days. |
Protect from light. Keep in original packaging when possible. Do not freeze reconstituted solution. Discard cloudy or discolored solutions.
Tesamorelin Protocol Mistakes & Troubleshooting
- 01
Cloudy or particulate solution
Discard the vial. Reconstituted tesamorelin should be clear and colorless. Cloudiness or visible particles can mean the powder did not fully dissolve, the vial was contaminated, or it was stored improperly.
- 02
Missed a dose
Skip the missed dose. Resume the next scheduled day at the normal time. Do not double up.
- 03
Burning or stinging at the injection site
This usually means the solution is too cold (just out of the fridge) or injected too quickly. Let the syringe sit at room temperature for a minute and inject slowly. Rotate sites every day.
- 04
Vial seems to be running out faster than expected
Recheck your reconstitution math. A 10 mg vial with 2.0 mL BAC water at 2 mg/day = 5 doses per vial. If you are getting fewer doses than expected, you may be drawing more than your target volume.
- 05
Powder is hard to dissolve
Roll the vial gently between your palms for 60 to 90 seconds. Do not shake. If powder is still stuck on the wall, gently swirl. Avoid forcing the water against the powder.
- 06
Blood sugar rising on tesamorelin
Bring this to a clinician. The label recommends blood-sugar monitoring before and during use. Persistently high blood sugar may need dose review or stopping.
- 07
Severe reaction or persistent side effect
Stop tesamorelin and seek qualified medical care. Severe allergic reactions, persistent edema, or signs of high IGF-1 (joint pain, carpal tunnel, headaches) warrant clinical review.
Tesamorelin Regulatory Status
As of May 2026, tesamorelin (brand name Egrifta) is FDA-approved for reduction of excess abdominal fat in HIV-infected adults with lipodystrophy. It was first approved in November 2010. Egrifta SV (2 mg vial) and Egrifta WR (11.6 mg weekly-reconstitution vial) are the current commercial products. The original 1 mg vial format has been discontinued.
Tesamorelin is not a scheduled controlled substance. It is a prescription drug for its approved use. Use for non-HIV indications — including off-label use for general visceral fat reduction, metabolic syndrome, or body composition — is not FDA-approved and is considered off-label or research use.
Research-grade tesamorelin sold by grey-market peptide suppliers is not approved for human consumption. These products are labeled for research use only.
Tesamorelin is also a prohibited substance under WADA (World Anti-Doping Agency) rules as a peptide hormone in class S2.
Tesamorelin vs Sermorelin vs CJC-1295
Tesamorelin, sermorelin, and CJC-1295 all hit the same target (the GHRH receptor) but differ in important ways. The biggest gaps: tesamorelin is the only one with FDA approval and Phase III data showing visceral fat reduction. Sermorelin has the longest history of clinical use but is no longer made as a prescription product. CJC-1295 (with DAC) needs the fewest injections (weekly versus daily) but has no comparable trial evidence.
GHRH Peptides — Side-by-Side
Feature
Receptor Target
Tesamorelin
GHRH receptor
Sermorelin
GHRH receptor
CJC-1295 (DAC)
GHRH receptor
Feature
Peptide Length
Tesamorelin
44 amino acids (modified)
Sermorelin
29 amino acids
CJC-1295 (DAC)
29 amino acids + DAC
Feature
Half-Life
Tesamorelin
26-38 minutes
Sermorelin
11-12 minutes
CJC-1295 (DAC)
~6-8 days
Feature
Dosing Frequency
Tesamorelin
Once daily
Sermorelin
Once nightly
CJC-1295 (DAC)
1-2x weekly
Feature
FDA Status
Tesamorelin
Approved (2010; Egrifta)
Sermorelin
Previously approved; discontinued
CJC-1295 (DAC)
Not FDA-approved
Feature
Phase III Trial Data
Tesamorelin
Yes (n=816 pooled)
Sermorelin
No adult Phase III VAT datasets
CJC-1295 (DAC)
No
Feature
Visceral Fat Reduction
Tesamorelin
15-20% at 26 weeks
Sermorelin
Not formally studied in RCTs
CJC-1295 (DAC)
Not formally studied in RCTs
Feature
Liver Fat Reduction
Tesamorelin
32% at 12 months (NAFLD trial)
Sermorelin
Not studied
CJC-1295 (DAC)
Not studied
Feature
Typical Research Dose
Tesamorelin
2 mg daily
Sermorelin
200-300 mcg nightly
CJC-1295 (DAC)
1-2 mg weekly
Feature
Unique Advantage
Tesamorelin
Only FDA-approved GHRH with deepest trial evidence
Sermorelin
Longest historical track record
CJC-1295 (DAC)
Weekly dosing convenience
| Feature | Tesamorelin | Sermorelin | CJC-1295 (DAC) |
|---|---|---|---|
| Receptor Target | GHRH receptor | GHRH receptor | GHRH receptor |
| Peptide Length | 44 amino acids (modified) | 29 amino acids | 29 amino acids + DAC |
| Half-Life | 26-38 minutes | 11-12 minutes | ~6-8 days |
| Dosing Frequency | Once daily | Once nightly | 1-2x weekly |
| FDA Status | Approved (2010; Egrifta) | Previously approved; discontinued | Not FDA-approved |
| Phase III Trial Data | Yes (n=816 pooled) | No adult Phase III VAT datasets | No |
| Visceral Fat Reduction | 15-20% at 26 weeks | Not formally studied in RCTs | Not formally studied in RCTs |
| Liver Fat Reduction | 32% at 12 months (NAFLD trial) | Not studied | Not studied |
| Typical Research Dose | 2 mg daily | 200-300 mcg nightly | 1-2 mg weekly |
| Unique Advantage | Only FDA-approved GHRH with deepest trial evidence | Longest historical track record | Weekly dosing convenience |
Tesamorelin is the only GHRH peptide with active FDA approval and validated Phase III visceral-fat data.
For a metabolic angle outside the GHRH axis, see the SLU-PP-332 protocol — a pan-ERR exercise-mimetic small molecule, not a GH-releasing peptide. For compound-specific deep dives, see the Sermorelin protocol and CJC-1295 (DAC) protocol.
Tesamorelin Blood Tests & Monitoring
Tesamorelin is an approved GHRH analog in a specific clinical context and increases IGF-1. Monitoring focuses on IGF-1, glucose control, diabetes/retinopathy context, lipids, and GH-axis symptoms.
Blood test markers to discuss with a clinician
Marker
IGF-1
Why it matters
Tesamorelin increases IGF-1, so this marker helps track downstream GH-axis exposure.
Timing
Follow-up
Marker
A1c
Why it matters
Reviews longer-term glucose control because tesamorelin can affect glucose tolerance.
Timing
Baseline
Marker
Fasting glucose
Why it matters
Gives a current glucose snapshot during GH-axis stimulation.
Timing
Follow-up
Marker
Lipid panel
Why it matters
Tracks cardiometabolic changes that may be part of tesamorelin-related monitoring.
Timing
Follow-up
Marker
TSH and free T4
Why it matters
Adds thyroid context because thyroid status can affect energy, weight, and GH-axis interpretation.
Timing
Optional
| Marker | Why it matters | Timing |
|---|---|---|
| IGF-1 | Tesamorelin increases IGF-1, so this marker helps track downstream GH-axis exposure. | Follow-up |
| A1c | Reviews longer-term glucose control because tesamorelin can affect glucose tolerance. | Baseline |
| Fasting glucose | Gives a current glucose snapshot during GH-axis stimulation. | Follow-up |
| Lipid panel | Tracks cardiometabolic changes that may be part of tesamorelin-related monitoring. | Follow-up |
| TSH and free T4 | Adds thyroid context because thyroid status can affect energy, weight, and GH-axis interpretation. | Optional |
Monitoring guidance is label-informed for tesamorelin, especially IGF-1 and glucose-related monitoring.
At-home blood test option
Easy at home option to monitor core metrics during research cycles.

Partner link: PDP may earn a commission at no cost to you.
Simple timing framework
Baseline
Discuss baseline labs before starting, especially with diabetes, retinopathy, sleep apnea, edema, cancer history, thyroid disease, or glucose intolerance.
Follow-up
Re-check IGF-1 and glucose markers after 6-12 weeks or after meaningful dose changes.
Longer term
For longer protocols, review IGF-1 and metabolic trends periodically with a clinician.
How to interpret the labs
- IGF-1 monitoring is especially relevant because tesamorelin labeling discusses IGF-1 elevation.
- Diabetes, glucose intolerance, and retinopathy history need clinician-guided review.
- Cancer history or active malignancy concerns need medical review before GH-axis stimulation is considered.
Do not wait for routine labs
Vision changes, rapid swelling, severe headaches, chest pain, shortness of breath, or allergic symptoms need medical review. New numbness, wrist pain, or worsening sleep apnea symptoms should be discussed with a clinician.
FAQ
Q1: What is the starting dose of tesamorelin?
Tesamorelin starts at full dose from day one — no ramp-up. The dose depends on the formulation: 1.4 mg daily for Egrifta SV, 1.28 mg daily for Egrifta WR, or 2 mg daily for research vials (matching the original Phase III trial dose). Inject under the skin of the abdomen, rotating sites each day.
Q2: What is tesamorelin's half-life?
Tesamorelin's half-life is about 26 to 38 minutes. That means the compound itself clears from the bloodstream quickly. The growth hormone and IGF-1 it triggers continue working in the body well beyond that window, which is why once-daily dosing is effective despite the short half-life.
Q3: Is tesamorelin FDA-approved?
Yes. Tesamorelin (brand name Egrifta) was FDA-approved in November 2010 for reduction of excess abdominal fat in HIV-infected adults with lipodystrophy. It is the only FDA-approved GHRH analogue. Use for any other purpose is off-label.
Q4: What are the most common side effects of tesamorelin?
The most common side effects are injection-site reactions (redness, itching, swelling), fluid retention with swelling in hands or feet, joint pain, and muscle aches. Some people see changes in blood sugar levels, so periodic monitoring is recommended. About half of trial participants developed antibodies to tesamorelin, but this did not reduce the drug's fat-reduction effects.
Q5: How do you reconstitute tesamorelin?
For research vials, add bacteriostatic water slowly down the inside wall of the vial (typically 2.0 mL for a 10 mg vial). Roll gently to dissolve. Do not shake. Refrigerate at 2-8C. For exact volumes by vial size, see the reconstitution table above or use the Peptide Reconstitution Calculator.
Q6: How much bacteriostatic water should be added to tesamorelin?
Common setups: 0.5 to 1.0 mL for a 2 mg vial, 1.0 to 2.5 mL for a 5 mg vial, and 2.0 to 5.0 mL for a 10 mg vial. Less water means a stronger mix and smaller injections. More water means a weaker mix and easier-to-measure injections. Use the reconstitution calculator for exact unit math.
Q7: What results can be expected from tesamorelin?
Phase III trials showed visceral fat reduction of about 12-20% over 26 weeks of daily 2 mg dosing. A 12-month NAFLD trial reported about 32% liver-fat reduction versus placebo. When trial participants stopped tesamorelin, belly fat trended back toward pre-treatment levels, which suggests continued use is needed to keep results. Personal results vary and are not guaranteed.
Q8: How does tesamorelin compare to sermorelin and CJC-1295?
All three target the GHRH receptor. Tesamorelin is the only one with current FDA approval and Phase III trial data showing visceral fat reduction. Sermorelin has the longest history of clinical use but is no longer made as a prescription product. CJC-1295 (with DAC) lasts longer (weekly dosing) but has no comparable trial evidence. See the Sermorelin protocol and CJC-1295 DAC protocol for compound-specific guides.
Q9: What vial sizes does tesamorelin come in?
FDA-approved products are Egrifta SV (2 mg vial) and Egrifta WR (11.6 mg vial). Research-grade vials commonly come in 2 mg, 5 mg, and 10 mg sizes. The legacy 1 mg Egrifta vial is discontinued.
Q10: How many doses are in a 10 mg vial of tesamorelin?
At 2 mg per day, a 10 mg vial gives about 5 daily doses. Reconstitute with 2.0 mL of bacteriostatic water for a 5,000 mcg/mL solution. Each daily dose is 0.40 mL (40 units on a U-100 insulin syringe). Round up your vial count to allow for priming losses.
Q11: What is the best time of day for tesamorelin?
Tesamorelin can be injected at any time of day. The original Phase III trials did not require bedtime dosing. Some users prefer morning to align with daytime activity; others prefer bedtime to match the body's natural overnight GH pulse. Consistency matters more than timing.
Q12: How should reconstituted tesamorelin be stored?
Storage depends on the formulation. Egrifta SV is single-use and should be injected right after mixing per the label. Egrifta WR and research-grade reconstituted vials are stored refrigerated at 35.6 to 46.4F (2-8C) for up to 28 days. Do not freeze reconstituted solution. Discard if cloudy or discolored.
Q13: Is tesamorelin a controlled or scheduled drug?
No. Tesamorelin is not a scheduled controlled substance under the U.S. Controlled Substances Act. It is a prescription drug for its FDA-approved use in HIV lipodystrophy. It is, however, a prohibited substance under WADA anti-doping rules (class S2 peptide hormone).
Q14: What clinical trials have been done on tesamorelin?
Tesamorelin has two Phase III lipodystrophy trials (LIPO-010 and CTR-1011), pooled analyses across 816 participants, the Stanley 2019 Lancet HIV NAFLD trial, the Stanley 2014 JAMA abdominal-fat trial, and mechanistic transcriptomic and proteomic follow-up papers. An ongoing registered study (ClinicalTrials.gov NCT02196831) continues to evaluate these outcomes.
Q15: Is titration necessary for tesamorelin?
No. The FDA label and Phase III trials both start patients at the full dose on day one. Tesamorelin does not need the slow ramp-up that many other peptides use. This is one of the things that makes it different from community-derived peptide protocols.
Q16: Where can I calculate reconstitution and syringe units?
Use the PepPal calculator for exact dose-to-unit conversions across any vial size and BAC water volume.
Sources & Research
- 1. Falutz J, et al. Effects of tesamorelin (TH9507), a growth hormone-releasing factor analog, in HIV-infected patients with excess abdominal fat: pooled analysis of two multicenter Phase 3 trials with safety extension data. Journal of Clinical Endocrinology & Metabolism (2010)
- 2. Stanley TL, Fourman LT, Feldpausch MN, et al. Effects of tesamorelin on non-alcoholic fatty liver disease in HIV: a randomised, double-blind, multicentre trial. Lancet HIV (2019)
- 3. Fourman LT, Stanley TL, et al. Effects of tesamorelin on hepatic transcriptomic signatures in HIV-associated NAFLD. JCI Insight (2020)
- 4. Fourman LT, Stanley TL, et al. Delineating tesamorelin response pathways in HIV-associated NAFLD using a targeted proteomic and transcriptomic approach. Scientific Reports (2021)
- 5. Grinspoon S, et al. Effects of a Growth Hormone-Releasing Hormone Analog on Endogenous GH Pulsatility and Insulin Sensitivity in Healthy Men. Journal of Clinical Endocrinology & Metabolism (2011)
- 6. Ishida J, et al. Growth hormone secretagogues: history, mechanism of action, and clinical development. JCSM Rapid Communications (2020)
- 7. U.S. Food and Drug Administration Egrifta SV (tesamorelin) prescribing information. FDA (2024)
- 8. U.S. Food and Drug Administration Egrifta WR (tesamorelin) prescribing information. FDA (2025)
- 9. Drugs.com Tesamorelin Monograph for Professionals. Drugs.com (2025)
- 10. National Institutes of Health LiverTox: Tesamorelin. NCBI Bookshelf (2023)
- 11. ClinicalTrials.gov Tesamorelin Effects on Liver Fat and Histology in HIV (NCT02196831). ClinicalTrials.gov (2024)
- 12. Falutz J, McLaughlin T, et al. Efficacy and safety of tesamorelin in people with HIV on integrase inhibitors. AIDS (2024)
Related Dosing Protocols
Written by Garret Grant
Founder & Lead Researcher · B.S. Civil Engineering, UCLA
Last updated: May 2026
Human-researched and AI-assisted with full editorial review. I verify sources, protocol interpretation, and final judgments personally. See methodology.
Share this page
