Sermorelin Quick Start
Sermorelin is a small peptide that copies the first 29 amino acids of the body's natural growth hormone-releasing hormone (GHRH). It is also called GRF 1-29 or sermorelin acetate. The short copy still fits the pituitary gland's GHRH receptor, so it can trigger a normal growth hormone (GH) pulse.
Most research-context protocols use 200-300 mcg once a night, by subcutaneous injection, about 30-60 minutes before bed on an empty stomach. This is meant to ride along with the body's largest natural GH pulse, which happens during early sleep.
Sermorelin has an unusual history. It was FDA-approved in 1990 as a diagnostic agent and again in 1997 as a treatment for childhood growth hormone deficiency (brand name Geref). Serono Laboratories pulled it from the market in 2008 for manufacturing reasons, not safety reasons. As of May 2026, sermorelin still sits in FDA 503A Category 1, which means licensed compounding pharmacies can legally prepare it. That is different from peptides like BPC-157 or CJC-1295, which sit on Category 2 and cannot be compounded.
Common dose
200-300 mcg subQ nightly. Titration usually starts at 100 mcg.
Half-life
About 11-12 minutes. That is why it is given nightly, not weekly.
Reconstitution
3 mL bacteriostatic (BAC) water in a 10 mg vial gives 3 units per 100 mcg on a U-100 insulin syringe.
Timing
30-60 minutes before sleep, on an empty stomach. Insulin and meals blunt the GH pulse.
Regulatory status
503A Category 1 in May 2026 - still legally compoundable. Branded Geref discontinued 2008.
Educational research reference only
This page is an educational research reference, not medical advice or a treatment plan. Sermorelin has historical FDA approvals but no current branded product. Work with a licensed clinician and a 503A or 503B compounding pharmacy if you are exploring sermorelin in a clinical setting.
Sermorelin Dosing Protocol & Schedule
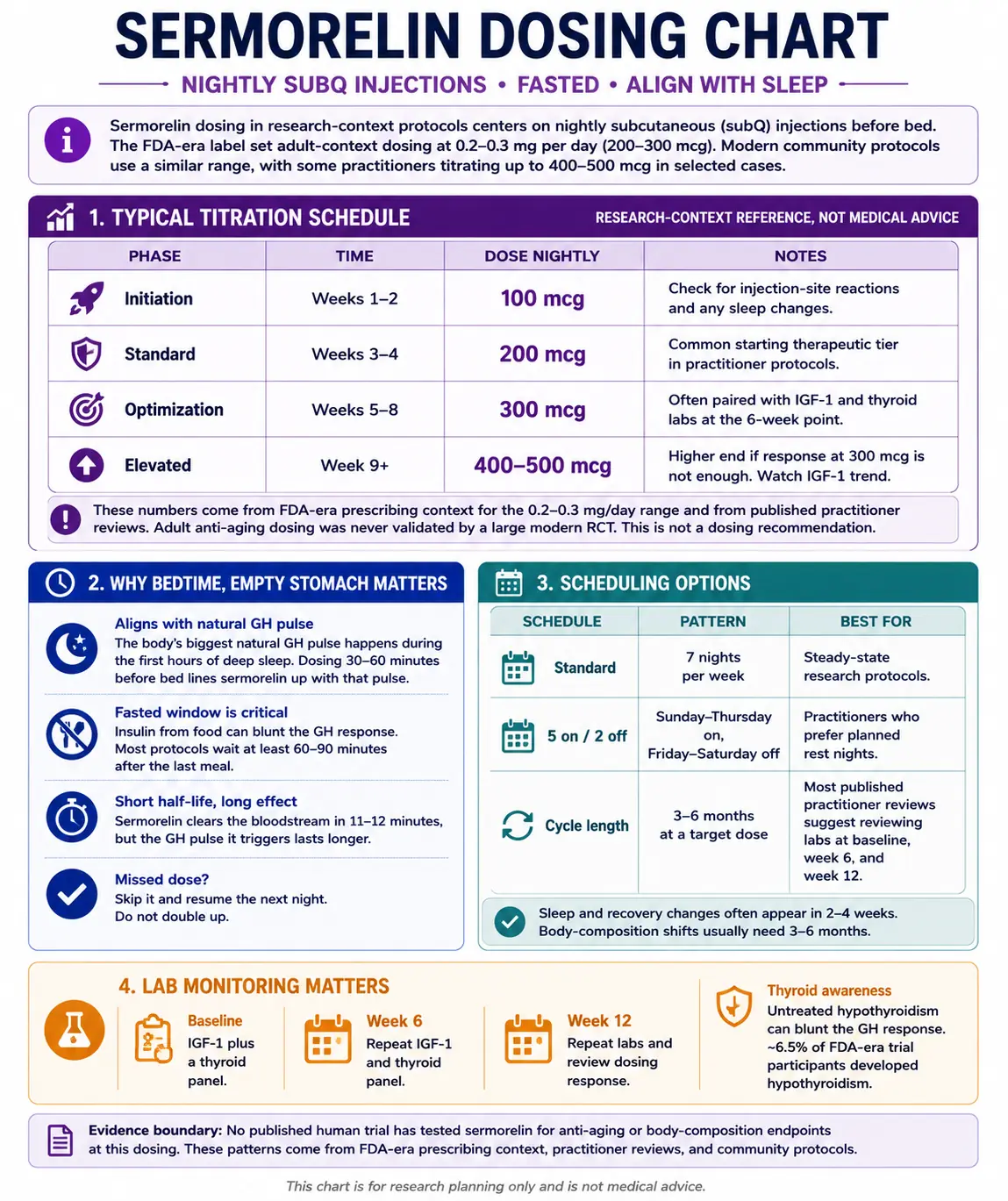
Sermorelin dosing in research-context protocols centers on nightly subcutaneous (subQ) injections before bed. The FDA-era label set adult-context dosing at 0.2-0.3 mg per day (200-300 mcg). Modern community protocols use a similar range, with some practitioners titrating up to 400-500 mcg in selected cases.
Higher doses do not always produce a bigger GH pulse. The body's natural "off switch" hormone, somatostatin, kicks in when GH rises too high. That feedback caps how much GH a single sermorelin dose can release. This is one reason sermorelin is considered lower-risk than direct GH injections.
Typical Titration Schedule
Sermorelin titration framework (research-context reference, not medical advice)
Phase
Initiation
Time
Weeks 1-2
Dose nightly
100 mcg
Notes
Check for injection-site reactions and any sleep changes.
Phase
Standard
Time
Weeks 3-4
Dose nightly
200 mcg
Notes
Common starting therapeutic tier in practitioner protocols.
Phase
Optimization
Time
Weeks 5-8
Dose nightly
300 mcg
Notes
Often paired with IGF-1 and thyroid labs at the 6-week point.
Phase
Elevated
Time
Week 9+
Dose nightly
400-500 mcg
Notes
Higher end if response at 300 mcg is not enough. Watch IGF-1 trend.
| Phase | Time | Dose nightly | Notes |
|---|---|---|---|
| Initiation | Weeks 1-2 | 100 mcg | Check for injection-site reactions and any sleep changes. |
| Standard | Weeks 3-4 | 200 mcg | Common starting therapeutic tier in practitioner protocols. |
| Optimization | Weeks 5-8 | 300 mcg | Often paired with IGF-1 and thyroid labs at the 6-week point. |
| Elevated | Week 9+ | 400-500 mcg | Higher end if response at 300 mcg is not enough. Watch IGF-1 trend. |
These numbers come from FDA-era prescribing context for the 0.2-0.3 mg/day range and from published practitioner reviews. Adult anti-aging dosing was never validated by a large modern RCT. This is not a dosing recommendation.
Why Bedtime, Empty Stomach Matters
- The body's biggest natural GH pulse happens during the first hours of deep sleep. Dosing 30-60 minutes before bed lines sermorelin up with that pulse.
- Insulin from food can blunt the GH response. Most protocols wait at least 60-90 minutes after the last meal.
- Sermorelin clears the bloodstream in 11-12 minutes, but the GH pulse it triggers lasts longer.
Scheduling Options
Common nightly scheduling models
Schedule
Standard
Pattern
7 nights per week
Best for
Steady-state research protocols.
Schedule
5 on / 2 off
Pattern
Sunday-Thursday on, Friday-Saturday off
Best for
Practitioners who prefer planned rest nights.
Schedule
Cycle length
Pattern
3-6 months at a target dose
Best for
Most published practitioner reviews suggest reviewing labs at baseline, week 6, and week 12.
| Schedule | Pattern | Best for |
|---|---|---|
| Standard | 7 nights per week | Steady-state research protocols. |
| 5 on / 2 off | Sunday-Thursday on, Friday-Saturday off | Practitioners who prefer planned rest nights. |
| Cycle length | 3-6 months at a target dose | Most published practitioner reviews suggest reviewing labs at baseline, week 6, and week 12. |
Sleep and recovery changes often appear in 2-4 weeks. Body-composition shifts usually need 3-6 months. If a dose is missed, skip it and resume the next night - do not double up.
Lab monitoring matters
Standard practice is baseline labs (IGF-1 plus a thyroid panel), repeat at week 6, and again at week 12. Untreated hypothyroidism can blunt the GH response. About 6.5% of FDA-era trial participants developed hypothyroidism, which is why thyroid checks were built into the original prescribing pattern.
Sermorelin Reconstitution Guide
Reconstitution is just adding sterile bacteriostatic (BAC) water to the dry sermorelin powder so it can be drawn into a syringe. The math is the same for any peptide: divide the total peptide amount by the BAC water volume to get the concentration in mcg/mL, then divide your target dose by that concentration to get the draw volume in mL.
Default 10 mg Vial Setup
With 10 mg of sermorelin and 3 mL of BAC water, the concentration is 3,333 mcg/mL. On a U-100 insulin syringe (where 100 units equals 1 mL), the math comes out to clean whole units:
10 mg sermorelin vial reconstituted with 3 mL BAC water (3,333 mcg/mL)
Target dose
100 mcg
Draw volume
0.03 mL
Units (U-100)
3 units
Target dose
200 mcg
Draw volume
0.06 mL
Units (U-100)
6 units
Target dose
300 mcg
Draw volume
0.09 mL
Units (U-100)
9 units
Target dose
400 mcg
Draw volume
0.12 mL
Units (U-100)
12 units
Target dose
500 mcg
Draw volume
0.15 mL
Units (U-100)
15 units
| Target dose | Draw volume | Units (U-100) |
|---|---|---|
| 100 mcg | 0.03 mL | 3 units |
| 200 mcg | 0.06 mL | 6 units |
| 300 mcg | 0.09 mL | 9 units |
| 400 mcg | 0.12 mL | 12 units |
| 500 mcg | 0.15 mL | 15 units |
Round to the nearest unit if your syringe does not show half-unit marks. For a different vial size or water volume, recheck with the calculator.
Other Common Vial Sizes
Other sermorelin vial sizes and BAC water amounts
Vial size
2 mg
BAC water
1 mL
Concentration
2,000 mcg/mL
200 mcg =
10 units
Vial size
5 mg
BAC water
2 mL
Concentration
2,500 mcg/mL
200 mcg =
8 units
Vial size
9 mg
BAC water
3 mL
Concentration
3,000 mcg/mL
200 mcg =
~6.7 units
Vial size
15 mg
BAC water
5 mL
Concentration
3,000 mcg/mL
200 mcg =
~6.7 units
| Vial size | BAC water | Concentration | 200 mcg = |
|---|---|---|---|
| 2 mg | 1 mL | 2,000 mcg/mL | 10 units |
| 5 mg | 2 mL | 2,500 mcg/mL | 8 units |
| 9 mg | 3 mL | 3,000 mcg/mL | ~6.7 units |
| 15 mg | 5 mL | 3,000 mcg/mL | ~6.7 units |
Smaller vials (2 mg, 5 mg) give the cleanest whole-unit numbers. Larger vials (9 mg, 15 mg) give the longest shelf life from one bottle of powder.
Step-by-Step Reconstitution
- 01
Wipe the vial stopper
Remove the plastic cap. Wipe the rubber stopper with an alcohol swab and let it dry.
- 02
Draw BAC water
Use a sterile syringe to draw the planned amount of bacteriostatic water (3 mL for a 10 mg vial).
- 03
Inject against the wall
Insert the needle at an angle and aim the water at the inside wall of the vial - not directly onto the powder.
- 04
Let it run down
Let the water flow gently down the side of the vial. Do not spray or force it.
- 05
Mix slowly
Remove the syringe. Roll the vial between your palms for 30-60 seconds until the powder fully dissolves. Do not shake.
- 06
Inspect
The solution should be clear and colorless. Discard if it looks cloudy, discolored, or has particles.
- 07
Label and refrigerate
Store upright at 2-8°C (36-46°F). Write the reconstitution date and the concentration on the label.
Need exact units for a custom setup?
If you have a non-standard vial size or different BAC water volume, use the free PepPal calculator to get the exact draw volume and syringe units.
Sermorelin Dosage Chart
This sermorelin dosage chart summarizes the common nightly titration schedule from 100 mcg up to 500 mcg, with dose escalation shown by week range.
How Sermorelin Works
Sermorelin works by talking to the pituitary gland, a small gland at the base of the brain that makes growth hormone (GH). The pituitary normally listens for a signal from the brain called GHRH (growth hormone-releasing hormone). When GHRH shows up, the pituitary releases a burst of GH.
Sermorelin is a short copy of GHRH. The natural hormone is 44 amino acids long. Sermorelin uses only the first 29. Despite being shorter, the 29-amino-acid version still fits the GHRH receptor and still triggers a GH pulse. That is the main mechanism.
The Built-in Safety Brake
The body has a natural "off switch" hormone called somatostatin. When GH levels climb too high, somatostatin steps in and quiets the pituitary back down. Because sermorelin works through the body's own GHRH pathway, this off switch stays active. GH levels rise in natural pulses rather than staying flat and elevated.
This is the key difference from direct GH injections. With direct GH, the dose you inject is the dose you get - the off switch is bypassed. With sermorelin, the brake is still on, so the body protects itself from very high GH levels even at higher sermorelin doses.
Pulsatile Release Matters
Healthy growth hormone in the body is pulsatile - it rises and falls in waves, with the biggest waves during early sleep. Sermorelin's short half-life (11-12 minutes) actually fits this pattern well. It nudges the pituitary into making a pulse, then clears out fast, leaving the body's own rhythm in place.
- Receptor: GHRH receptor on pituitary somatotroph cells.
- Trigger: cAMP signaling inside the cell, which causes GH release.
- Brake: Somatostatin feedback keeps GH from climbing too high.
- Why nightly: Sermorelin clears in minutes, but the GH pulse it triggers fits the natural early-night surge.
Sermorelin Supplies Needed
Affiliate disclosure: PDP may earn a commission when you use eligible supplier links, at no extra cost to you. The math below assumes a default 10 mg sermorelin vial, 3 mL BAC water per vial, and 200 mcg per night.
Recommended Supply
Use discount code PEPPAL at eligible peptide supplier checkouts.

Sermorelin Supply

SiPhox Health At-Home Blood Test
Injection Supplies
Disclosure: supply links may earn PDP a commission at no cost to you.
Sermorelin Vials (10 mg default)
One 10 mg vial holds 50 doses of 200 mcg. Once reconstituted, the solution should be used within 28 days, so longer cycles need fresh vials roughly once a month.
| Cycle length | Planning note |
|---|---|
4 weeks 1 vial | 28 nights of 200 mcg from one vial. |
8 weeks 2 vials | Start the second vial around day 28 to respect the 28-day reconstituted limit. |
12 weeks 3 vials | One vial per ~28 days of dosing. |
6 months 6 vials | Six vials cover ~168 nightly doses across a 24-week cycle. |
4 weeks
1 vial
28 nights of 200 mcg from one vial.
8 weeks
2 vials
Start the second vial around day 28 to respect the 28-day reconstituted limit.
12 weeks
3 vials
One vial per ~28 days of dosing.
6 months
6 vials
Six vials cover ~168 nightly doses across a 24-week cycle.
Insulin Syringes (U-100)
Use 0.3 mL / 30-unit syringes. With 3 mL BAC water, 200 mcg equals 6 units - easy to read on a U-100 syringe.
| Cycle length | Planning note |
|---|---|
4 weeks 28-30 syringes | One syringe per nightly injection. Round up for occasional losses. |
8 weeks 60 syringes | One syringe per nightly injection. |
12 weeks 90 syringes | One syringe per nightly injection. |
6 months 180 syringes | About one 100-count box plus a partial second box. |
4 weeks
28-30 syringes
One syringe per nightly injection. Round up for occasional losses.
8 weeks
60 syringes
One syringe per nightly injection.
12 weeks
90 syringes
One syringe per nightly injection.
6 months
180 syringes
About one 100-count box plus a partial second box.
Bacteriostatic Water
Use 3 mL per 10 mg vial. Most BAC water bottles are 10 mL or 30 mL.
| Cycle length | Planning note |
|---|---|
4-12 weeks 1 x 10 mL bottle | 4 weeks: One vial uses 3 mL; one bottle gives margin.; 8 weeks: Two vials use 6 mL total; one bottle is enough.; 12 weeks: Three vials use 9 mL; one 10 mL bottle gives a little margin. |
6 months 2 x 10 mL bottles | Six vials use 18 mL; two bottles cover the cycle with margin. |
4-12 weeks
1 x 10 mL bottle
4 weeks: One vial uses 3 mL; one bottle gives margin.; 8 weeks: Two vials use 6 mL total; one bottle is enough.; 12 weeks: Three vials use 9 mL; one 10 mL bottle gives a little margin.
6 months
2 x 10 mL bottles
Six vials use 18 mL; two bottles cover the cycle with margin.
Round up for priming losses, dropped syringes, and protocol adjustments. If you use a 2 mg, 5 mg, 9 mg, or 15 mg vial instead, use the [PepPal calculator](https://www.peppal.app/calculator?compound=sermorelin) to recheck units.
Who Sermorelin Is For and Who Should Avoid It
Sermorelin is typically discussed in two settings: pediatric GH deficiency in the historical FDA-era label, and adult off-label use through 503A or 503B compounding pharmacies for GH optimization, sleep, and body composition. Both should involve a licensed clinician.
Who Generally Avoids Sermorelin
- Known allergy to sermorelin or any of the ingredients in the formulation.
- Pregnancy or breastfeeding. The FDA-era label flagged sermorelin as Pregnancy Category C. There are no controlled studies in pregnant women, and it is unknown whether sermorelin passes into breast milk.
- Active cancer. GH and IGF-1 can act as growth signals on existing cells. Most protocols screen for active malignancy before starting.
- Growth hormone deficiency from an intracranial lesion - the FDA-era label specifically said these patients were not studied and were not recommended for sermorelin therapy.
- Untreated hypothyroidism. This blunts the GH response and should be corrected first.
- On glucocorticoid therapy. Glucocorticoids can blunt the response to sermorelin per the FDA-era label drug interactions.
Clinical oversight
Sermorelin therapy was, in the FDA-era label, supposed to be carried out under the regular guidance of a physician experienced in growth-hormone-related conditions. That is still the standard expectation for compounded sermorelin in 2026.
Sermorelin Side Effects & Safety
Sermorelin has a published safety profile from its FDA-era pediatric clinical trial program. Across 350 patients in those trials, the peptide was generally well-tolerated. Most side effects were mild, local, and resolved on their own.
Common Side Effects
- Injection-site reactions (about 1 in 6 participants, roughly 17%): pain, swelling, or redness at the injection site. Usually mild, usually went away on their own. Only 3 of 350 patients stopped treatment because of them.
Less Common Side Effects (each under 1%)
- Headache
- Facial flushing
- Difficulty swallowing (dysphagia)
- Dizziness
- Restlessness or hyperactivity
- Drowsiness
- Changes in taste
- Hives (urticaria)
- Nausea or vomiting (more often with IV diagnostic use than nightly subQ)
Things to Monitor
- Antibody development. About 70% of FDA-era trial participants developed anti-GRF antibodies at least once during treatment. The label noted that these antibodies did not appear to meaningfully reduce sermorelin's effectiveness or cause specific reactions in most participants.
- Hypothyroidism. About 6.5% of trial participants developed hypothyroidism. Untreated hypothyroidism reduces the body's response to GH stimulation, so baseline and follow-up thyroid labs are standard practice.
- Allergic reactions. Like any injected peptide, sermorelin can cause local or, rarely, systemic allergic reactions. Stop and seek medical attention for swelling of the face, mouth, or throat, trouble breathing, or widespread hives.
Lower-Risk Profile Compared to Direct GH
Because sermorelin works through the body's own feedback system, the risk of pushing GH or IGF-1 too high is built-in lower than with direct recombinant GH injections. Clinical data also showed no significant abuse or dependence potential.
For a broader cross-class safety reference, see the PepPal Peptide Side Effects Guide.
Sermorelin Timeline & What to Monitor
Most published practitioner reviews and FDA-era clinical context line up on a similar timeline. Sleep and recovery feedback tend to show up first. Body-composition changes take much longer, partly because they depend on weeks of steady GH and IGF-1 elevation, not a single pulse.
Reasonable monitoring framework for a sermorelin protocol
Timepoint
Baseline
What to check
IGF-1, full thyroid panel, fasting glucose, comprehensive metabolic panel
Why
Establish starting values before therapy and rule out untreated hypothyroidism.
Timepoint
Weeks 2-4
What to check
Subjective sleep quality, recovery, injection-site tolerance
Why
Sleep changes are usually the first reported shift.
Timepoint
Week 6
What to check
Repeat IGF-1, thyroid panel
Why
First objective check on whether the protocol is producing a GH/IGF-1 response.
Timepoint
Week 12
What to check
Repeat IGF-1, thyroid panel, body-composition markers if relevant
Why
Mid-point on a 3-6 month cycle.
Timepoint
Month 6
What to check
Full re-check of baseline labs plus body composition
Why
Most modern practitioner protocols re-evaluate the cycle around this point.
| Timepoint | What to check | Why |
|---|---|---|
| Baseline | IGF-1, full thyroid panel, fasting glucose, comprehensive metabolic panel | Establish starting values before therapy and rule out untreated hypothyroidism. |
| Weeks 2-4 | Subjective sleep quality, recovery, injection-site tolerance | Sleep changes are usually the first reported shift. |
| Week 6 | Repeat IGF-1, thyroid panel | First objective check on whether the protocol is producing a GH/IGF-1 response. |
| Week 12 | Repeat IGF-1, thyroid panel, body-composition markers if relevant | Mid-point on a 3-6 month cycle. |
| Month 6 | Full re-check of baseline labs plus body composition | Most modern practitioner protocols re-evaluate the cycle around this point. |
These are reasonable lab markers based on the FDA-era label and published practitioner reviews. They are not a treatment plan. Your clinician decides what to order and when.
Sermorelin Clinical Evidence Context
Sermorelin has an unusual evidence profile for a research-context peptide. It was actually FDA-approved twice: once in 1990 as a diagnostic agent for GH deficiency, and again in 1997 as a treatment for childhood GH deficiency. The branded version (Geref) was pulled in 2008 for manufacturing reasons, not safety reasons.
So sermorelin has real FDA-era trial data - but most of it is in children. Adult use today rests on practitioner experience, published reviews, and pharmacokinetic (PK) studies, not large modern randomized controlled trials.
FDA-era diagnostic program (1990)
Single-dose IV sermorelin produced rapid, specific GH release in healthy children and a blunted response in GH-deficient children. This is the data that supported sermorelin's diagnostic FDA approval.
FDA-era pediatric treatment program (1997)
Children with growth hormone deficiency received nightly subQ sermorelin at 0.2-0.3 mg/day. Trials showed significant increases in GH release and growth velocity at 6 months, with efficacy reported through 36 months.
Pharmacokinetics (Ishida et al., 2020)
Pooled PK data showed a half-life of about 11-12 minutes after either IV or subQ dosing, plasma clearance of 2.4-2.8 L/min, and rapid time-to-peak after subcutaneous injection.
Adult-context review (Walker, 2006)
Walker's review in Clinical Interventions in Aging discussed sermorelin for adult GH insufficiency, highlighting the pituitary-supportive and feedback-regulated nature of GHRH stimulation versus direct GH replacement.
Modern adult RCT data
Large modern randomized controlled trials in adults are limited after the 2008 commercial discontinuation. Current adult dosing rests on practitioner experience and published reviews rather than fresh Phase 3 trials.
Bottom line: sermorelin has more historical clinical evidence than most peptides discussed in research-use forums, but modern adult evidence is thin. That gap is a real boundary on any specific dosing claim.
Sermorelin Storage & Handling
Sermorelin storage by form
Form
Lyophilized (powder)
Temperature
-4°F (-20°C)
Time
Long-term (years)
Form
Lyophilized (powder)
Temperature
36-46°F (2-8°C)
Time
Months
Form
Lyophilized (powder)
Temperature
59-77°F (15-25°C)
Time
Weeks (shipping tolerance)
Form
Reconstituted
Temperature
36-46°F (2-8°C)
Time
Up to 28 days
Form
Reconstituted (frozen)
Temperature
-4°F (-20°C)
Time
Not recommended
| Form | Temperature | Time |
|---|---|---|
| Lyophilized (powder) | -4°F (-20°C) | Long-term (years) |
| Lyophilized (powder) | 36-46°F (2-8°C) | Months |
| Lyophilized (powder) | 59-77°F (15-25°C) | Weeks (shipping tolerance) |
| Reconstituted | 36-46°F (2-8°C) | Up to 28 days |
| Reconstituted (frozen) | -4°F (-20°C) | Not recommended |
Protect from light. Use bacteriostatic water for multi-dose handling. Do not freeze reconstituted sermorelin. Discard if the solution is cloudy, has particles, or is more than 28 days past reconstitution.
Short room-temperature windows (for example during travel) are usually fine, but refrigerate again as soon as possible.
Sermorelin Protocol Mistakes & Troubleshooting
Common issues and what to do
Issue
Cloudy vial after mixing
What it means
The solution may be contaminated or the powder may not have fully dissolved. Cloudiness can also signal a damaged or expired product.
Common response
Do not use. Discard and start fresh.
Issue
Missed a nightly dose
What it means
One missed dose is not a crisis. The GH pulse is short and self-limiting.
Common response
Skip the missed dose and resume the next night. Do not double up.
Issue
Ate a large meal before injection
What it means
Insulin from the meal can blunt the GH pulse.
Common response
Wait 60-90 minutes after the last meal before injecting. Consistency matters more than perfection.
Issue
Used too much or too little BAC water
What it means
The concentration changes, so the syringe-unit math no longer matches the chart.
Common response
Use the PepPal calculator with your actual BAC water volume to get the corrected unit count.
Issue
Persistent injection-site redness
What it means
Most reactions are mild and resolve. Persistent or expanding redness can signal infection or a stronger allergic reaction.
Common response
Rotate injection sites. If redness expands or is painful, stop and contact a clinician.
Issue
No subjective change after 8 weeks
What it means
Response varies. Untreated hypothyroidism or poor sleep timing can blunt the protocol.
Common response
Recheck thyroid labs and IGF-1. Review timing (empty stomach, before bed). Discuss with your clinician.
| Issue | What it means | Common response |
|---|---|---|
| Cloudy vial after mixing | The solution may be contaminated or the powder may not have fully dissolved. Cloudiness can also signal a damaged or expired product. | Do not use. Discard and start fresh. |
| Missed a nightly dose | One missed dose is not a crisis. The GH pulse is short and self-limiting. | Skip the missed dose and resume the next night. Do not double up. |
| Ate a large meal before injection | Insulin from the meal can blunt the GH pulse. | Wait 60-90 minutes after the last meal before injecting. Consistency matters more than perfection. |
| Used too much or too little BAC water | The concentration changes, so the syringe-unit math no longer matches the chart. | Use the PepPal calculator with your actual BAC water volume to get the corrected unit count. |
| Persistent injection-site redness | Most reactions are mild and resolve. Persistent or expanding redness can signal infection or a stronger allergic reaction. | Rotate injection sites. If redness expands or is painful, stop and contact a clinician. |
| No subjective change after 8 weeks | Response varies. Untreated hypothyroidism or poor sleep timing can blunt the protocol. | Recheck thyroid labs and IGF-1. Review timing (empty stomach, before bed). Discuss with your clinician. |
These are reasonable troubleshooting steps, not a substitute for clinician guidance.
Sermorelin Regulatory Status
Sermorelin's regulatory status confuses a lot of people. Here is the timeline, as of May 2026:
Sermorelin regulatory timeline
Year
1990
Status
FDA-approved as a diagnostic agent for GH deficiency.
Year
1997
Status
FDA-approved as a treatment for childhood GH deficiency, brand name Geref, made by Serono Laboratories.
Year
2008
Status
Serono voluntarily discontinued the branded Geref product. Reason: manufacturing process difficulties, not safety concerns.
Year
2023-2024
Status
FDA moved 19 other peptides (BPC-157, CJC-1295, ipamorelin, TB-500, and others) from 503A Category 1 to Category 2. Sermorelin was not moved. It remained in Category 1.
Year
May 2026
Status
Sermorelin still sits in FDA 503A Category 1, meaning licensed compounding pharmacies can legally prepare it for patients with a valid prescription.
| Year | Status |
|---|---|
| 1990 | FDA-approved as a diagnostic agent for GH deficiency. |
| 1997 | FDA-approved as a treatment for childhood GH deficiency, brand name Geref, made by Serono Laboratories. |
| 2008 | Serono voluntarily discontinued the branded Geref product. Reason: manufacturing process difficulties, not safety concerns. |
| 2023-2024 | FDA moved 19 other peptides (BPC-157, CJC-1295, ipamorelin, TB-500, and others) from 503A Category 1 to Category 2. Sermorelin was not moved. It remained in Category 1. |
| May 2026 | Sermorelin still sits in FDA 503A Category 1, meaning licensed compounding pharmacies can legally prepare it for patients with a valid prescription. |
This is the most important practical difference between sermorelin and most other peptides in research-use discussions. Most research peptides cannot be legally compounded today. Sermorelin can.
Compounded vs research-use
Compounded sermorelin from a licensed 503A or 503B pharmacy with a prescription is different from "research use only" (RUO) peptide sold online. Compounded products are intended for human use under clinician supervision. RUO products carry disclaimers that they are not for human use, and the FDA has issued warning letters when sellers cross that line.
Sermorelin vs Tesamorelin vs CJC-1295
Sermorelin, tesamorelin, and CJC-1295 (DAC) all target the same GHRH receptor in the pituitary. They differ in how long they last in the body, how often you inject, and how much modern evidence supports them.
Side-by-side GHRH analogue comparison
Receptor
Sermorelin
GHRH receptor
Tesamorelin
GHRH receptor
CJC-1295 (DAC)
GHRH receptor
Length
Sermorelin
29 amino acids
Tesamorelin
44 amino acids (modified)
CJC-1295 (DAC)
29 amino acids + DAC
Half-life
Sermorelin
11-12 minutes
Tesamorelin
26-38 minutes
CJC-1295 (DAC)
~6-8 days
Dosing
Sermorelin
Nightly subQ
Tesamorelin
Daily subQ
CJC-1295 (DAC)
1-2x per week subQ
FDA status
Sermorelin
Previously approved; discontinued 2008; Category 1
Tesamorelin
FDA-approved (Egrifta) for HIV-associated lipodystrophy
CJC-1295 (DAC)
Not FDA-approved; Category 2
Key clinical evidence
Sermorelin
FDA-era pediatric trials + PK data
Tesamorelin
Phase 3 visceral fat data
CJC-1295 (DAC)
Phase 1 PK and GH/IGF-1 duration data
Typical research dose
Sermorelin
200-300 mcg nightly
Tesamorelin
2 mg daily
CJC-1295 (DAC)
1-2 mg weekly
Main use case
Sermorelin
GH optimization, sleep, recovery
Tesamorelin
Visceral fat / metabolic context
CJC-1295 (DAC)
Sustained GH/IGF-1 elevation
| Sermorelin | Tesamorelin | CJC-1295 (DAC) | |
|---|---|---|---|
| Receptor | GHRH receptor | GHRH receptor | GHRH receptor |
| Length | 29 amino acids | 44 amino acids (modified) | 29 amino acids + DAC |
| Half-life | 11-12 minutes | 26-38 minutes | ~6-8 days |
| Dosing | Nightly subQ | Daily subQ | 1-2x per week subQ |
| FDA status | Previously approved; discontinued 2008; Category 1 | FDA-approved (Egrifta) for HIV-associated lipodystrophy | Not FDA-approved; Category 2 |
| Key clinical evidence | FDA-era pediatric trials + PK data | Phase 3 visceral fat data | Phase 1 PK and GH/IGF-1 duration data |
| Typical research dose | 200-300 mcg nightly | 2 mg daily | 1-2 mg weekly |
| Main use case | GH optimization, sleep, recovery | Visceral fat / metabolic context | Sustained GH/IGF-1 elevation |
All three target the same receptor but are not interchangeable. Half-life, FDA status, and access pathway differ.
Quick Decision Framework
- Sermorelin has the longest historical track record and is currently the most accessible GHRH analogue via compounding pharmacies. Best fit when pulsatile, feedback-controlled GH is the goal.
- Tesamorelin has the strongest modern RCT evidence, specifically for visceral fat reduction in HIV-associated lipodystrophy. Also legally accessible as an FDA-approved drug.
- CJC-1295 (DAC) offers the longest dosing interval but sits in 503A Category 2, so it cannot be legally compounded in the U.S. today.
For full compound-specific guides, see the Tesamorelin protocol and CJC-1295 (DAC) protocol.
Sermorelin Blood Tests & Monitoring
Sermorelin is a GHRH analog that stimulates the GH-axis. Monitoring focuses on IGF-1, glucose handling, thyroid context, lipids, and fluid-retention symptoms.
Blood test markers to discuss with a clinician
Marker
IGF-1
Why it matters
Shows the downstream signal after GH-axis stimulation and helps frame pathway intensity.
Timing
Follow-up
Marker
A1c
Why it matters
Tracks longer-term glucose control because GH-axis changes may affect insulin sensitivity.
Timing
Baseline
Marker
Fasting glucose
Why it matters
Gives a current glucose snapshot before and during GH-axis protocols.
Timing
Follow-up
Marker
Lipid panel
Why it matters
Reviews cholesterol and triglyceride trends during body-composition or GH-axis changes.
Timing
Baseline
Marker
TSH and free T4
Why it matters
Thyroid status can affect energy, weight, and GH-axis interpretation.
Timing
Optional
| Marker | Why it matters | Timing |
|---|---|---|
| IGF-1 | Shows the downstream signal after GH-axis stimulation and helps frame pathway intensity. | Follow-up |
| A1c | Tracks longer-term glucose control because GH-axis changes may affect insulin sensitivity. | Baseline |
| Fasting glucose | Gives a current glucose snapshot before and during GH-axis protocols. | Follow-up |
| Lipid panel | Reviews cholesterol and triglyceride trends during body-composition or GH-axis changes. | Baseline |
| TSH and free T4 | Thyroid status can affect energy, weight, and GH-axis interpretation. | Optional |
Monitoring guidance is GHRH/GH-axis pathway-based, with IGF-1 used as the main downstream monitoring marker.
At-home blood test option
Easy at home option to monitor core metrics during research cycles.

Partner link: PDP may earn a commission at no cost to you.
Simple timing framework
Baseline
Discuss baseline labs before starting, especially with diabetes risk, sleep apnea, edema, thyroid disease, pituitary history, or cancer history.
Follow-up
Re-check IGF-1 and metabolic markers after 6-8 weeks or after meaningful dose changes.
Longer term
For longer protocols, review GH-axis and metabolic trends every 3-6 months with a clinician.
How to interpret the labs
- IGF-1 should be interpreted with age, symptoms, and baseline values.
- Fluid retention, joint pain, numbness, sleep apnea symptoms, and glucose changes matter during GH-axis protocols.
- Cancer history or active malignancy concerns require clinician-guided review before GH-axis stimulation is considered.
Do not wait for routine labs
Rapid swelling, severe headaches, vision changes, chest pain, or shortness of breath needs medical review. New numbness, wrist pain, or worsening sleep apnea symptoms should be discussed with a clinician.
FAQ
Q1: What is the starting dose of sermorelin?
Common adult research-context starting guidance is 100-200 mcg subQ at bedtime for the first 1-2 weeks, then titrating to 200-300 mcg nightly. The historical FDA-era pediatric label used 0.2-0.3 mg/day. Adult protocols are typically adjusted based on response, sleep changes, and IGF-1 lab trends. This is not a dosing recommendation - work with a licensed clinician.
Q2: How much BAC water do you use to reconstitute a 10 mg sermorelin vial?
A common research-context setup is 3 mL of bacteriostatic water in a 10 mg vial, which gives a 3,333 mcg/mL concentration. On a U-100 insulin syringe, that produces clean whole-unit doses: 100 mcg = 3 units, 200 mcg = 6 units, 300 mcg = 9 units. For other vial sizes or water volumes, recheck with the PepPal calculator.
Q3: How many units of sermorelin should I inject?
It depends on your vial size and how much BAC water you used. With the common 10 mg vial plus 3 mL BAC water setup, 200 mcg equals 6 units on a U-100 syringe and 300 mcg equals 9 units. For any other setup, use the calculator linked in this protocol to get the exact unit count.
Q4: What is sermorelin's half-life?
Sermorelin's plasma half-life is approximately 11-12 minutes after either IV or subQ injection, per Ishida et al. (2020) and the FDA-era prescribing data. Plasma clearance is in the 2.4-2.8 L/min range. The growth hormone pulse triggered by sermorelin lasts longer than the parent peptide stays in circulation.
Q5: Is sermorelin FDA-approved in 2026?
Sermorelin was FDA-approved in 1990 (diagnostic use) and 1997 (pediatric GH deficiency, brand name Geref). Serono Laboratories voluntarily discontinued Geref in 2008 due to manufacturing difficulties, not safety concerns. As of May 2026, there is no currently marketed FDA-approved sermorelin product. However, sermorelin still sits in FDA 503A Category 1, which means licensed compounding pharmacies can legally prepare it with a prescription.
Q6: Is sermorelin legal to compound in 2026?
Yes. Sermorelin remains in FDA 503A Category 1, which means licensed 503A and 503B compounding pharmacies can legally prepare it for patients with a valid prescription. This is different from most other research peptides (BPC-157, CJC-1295, ipamorelin, TB-500, and others), which sit in Category 2 and cannot be legally compounded.
Q7: What are the most common side effects of sermorelin?
Across 350 FDA-era trial patients, the most common side effect was a mild injection-site reaction - pain, swelling, or redness - in about 1 in 6 participants. Only 3 of 350 patients discontinued because of it. Less common effects (each under 1%) included headache, flushing, dizziness, difficulty swallowing, drowsiness, and hives. About 6.5% developed hypothyroidism during trials, which is why thyroid labs are standard monitoring.
Q8: What results can be expected from sermorelin?
Practitioner timelines and FDA-era data line up on a similar pattern: sleep and recovery changes often appear in 2-4 weeks. Body-composition shifts and IGF-1 increases usually need 3-6 months of consistent nightly dosing. Adult-population endpoint data outside practitioner literature is limited, so individual response varies.
Q9: How does sermorelin compare to tesamorelin and CJC-1295?
All three target the GHRH receptor, but they differ in half-life, dosing, and regulatory status. Sermorelin: ~12 minute half-life, nightly, 503A Category 1. Tesamorelin: ~26-38 minute half-life, daily, FDA-approved for HIV-associated lipodystrophy. CJC-1295 (DAC): ~6-8 day half-life, weekly, 503A Category 2 (cannot be legally compounded). See the Tesamorelin protocol and CJC-1295 (DAC) protocol for full details.
Q10: Should sermorelin be taken at night?
Most research-context protocols use sermorelin 30-60 minutes before bed on an empty stomach. The body's biggest natural growth hormone pulse happens during early sleep, and bedtime dosing rides along with that pulse. Eating raises insulin, which blunts the GH response, so most protocols wait 60-90 minutes after the last meal before injecting.
Q11: What vial sizes does sermorelin come in?
Common research-channel sermorelin vial sizes include 2 mg, 5 mg, 9 mg, 10 mg, and 15 mg lyophilized formats. The 10 mg vial is widely available from current compounded and research suppliers. Smaller vials (2 mg, 5 mg) give the cleanest whole-unit syringe math. Larger vials last longer once reconstituted - but remember the 28-day reconstituted-solution shelf life.
Q12: How should reconstituted sermorelin be stored?
Store reconstituted sermorelin upright in the refrigerator at 2-8°C (36-46°F), protect from light, and use within 28 days. Do not freeze reconstituted sermorelin. Discard if the solution is cloudy, discolored, or has any visible particles. Brief room-temperature windows (such as travel) are usually fine, but get it back into refrigeration as soon as possible.
Q13: Can sermorelin be stacked with ipamorelin?
Community protocols often discuss pairing sermorelin (a GHRH analogue) with ipamorelin (a GH secretagogue/GHRP) at bedtime to recruit both signaling pathways. Direct stack-specific clinical trial evidence is limited, and adding compounds increases the safety and quality-control surface area. Read the PepPal Peptide Stacking 101 guide before combining compounds.
Q14: Is this page medical advice?
No. This is an educational research reference. It summarizes published FDA-era data, pharmacokinetic literature, and practitioner-derived protocol structures so you have a clear starting point for a conversation with a licensed clinician. It is not a treatment plan and does not replace medical guidance.
Sources & Research
- 1. Ishida J, et al. Growth hormone secretagogues: history, mechanism of action, and clinical development. JCSM Rapid Communications (2020)
- 2. Walker RF. Sermorelin: A better approach to management of adult-onset growth hormone insufficiency? Clinical Interventions in Aging (2006)
- 3. Cunha JP. Sermorelin Acetate prescribing information. RxList (FDA-era label content, rev. 10/2/2001) (2022)
- 4. U.S. Food and Drug Administration. Certain Bulk Drug Substances for Use in Compounding that May Present Significant Safety Risks (503A Category 1 / Category 2 list). FDA.gov (2026)
- 5. U.S. Food and Drug Administration. July 23-24, 2026 Meeting of the Pharmacy Compounding Advisory Committee (503A Bulks List review). FDA.gov advisory committee calendar (2026)
- 6. Mayo Clinic. Sermorelin (injection route) - side effects and dosage. Mayo Clinic Drugs & Supplements (2025)
- 7. DrugBank. Sermorelin (DB00010) drug profile. DrugBank Online (2026)
- 8. PubChem. Sermorelin (CID 16132413). PubChem (NCBI) (2026)
- 9. Merriam GR, et al. Pharmacokinetics of growth hormone-releasing hormone(1-29)-NH2 and stimulation of growth hormone secretion in healthy subjects after intravenous or intranasal administration. Acta Paediatrica Supplement (1993)
- 10. Frier Levitt Attorneys at Law. Regulatory Status of Peptide Compounding in 2025 (sermorelin and NAD+ as Category 1 examples). Frier Levitt (2026)
- 11. ScienceDirect Topics. Sermorelin summary and references. ScienceDirect (Elsevier) (2025)
Related Dosing Protocols
Educational research reference
This page is an educational research reference, not medical advice or a treatment plan. Sermorelin was previously FDA-approved (Geref, discontinued 2008) and as of May 2026 remains legally compoundable through 503A and 503B pharmacies with a valid prescription. Always work with a licensed clinician for personal protocols.
Calculate vial math
Use the PepPal calculator for exact syringe units across any vial size and BAC water volume.
Open CalculatorWritten by Garret Grant
Founder & Lead Researcher · B.S. Civil Engineering, UCLA
Last updated: May 2026
Human-researched and AI-assisted with full editorial review. I verify sources, protocol interpretation, and final judgments personally. See methodology.
Share this page
