Ipamorelin Quick Start
Ipamorelin is a small research peptide. It tells the body to release its own growth hormone in short, natural pulses. It is sometimes called NNC 26-0161. Novo Nordisk first studied it in the late 1990s.
Most research protocols use 100 to 300 mcg per shot, one to three times per day. The shot goes under the skin (subcutaneous). Ipamorelin has a short half-life of about two hours, so each shot creates one clean GH pulse and then fades.
What makes ipamorelin stand out is how clean it is. Other peptides in its family (GHRP-2, GHRP-6) also raise cortisol, prolactin, and other hormones you do not want raised. Ipamorelin does not do that, even at high doses. That is why most research protocols pick it first.
Route
Subcutaneous shot only. Most protocols inject in the lower belly, thigh, or upper arm.
Dose range
100 to 300 mcg per shot. Past 300 mcg, GH release does not climb meaningfully.
Schedule
One to three shots per day. Bedtime is the most common single dose.
Fasting
Inject on an empty stomach. Food (especially carbs) blunts the GH pulse through insulin.
Cycle
Most community protocols run 8 to 12 weeks on, then 4 weeks off.
Status
Not FDA-approved. Banned in sport (WADA). Research-use only.
Disclaimer
This page is an educational research reference. It is not medical advice and not a treatment plan. Ipamorelin is not FDA-approved for any human use.
Need exact syringe units for your vial? Use the Pep Pal calculator to lock in the draw volume.
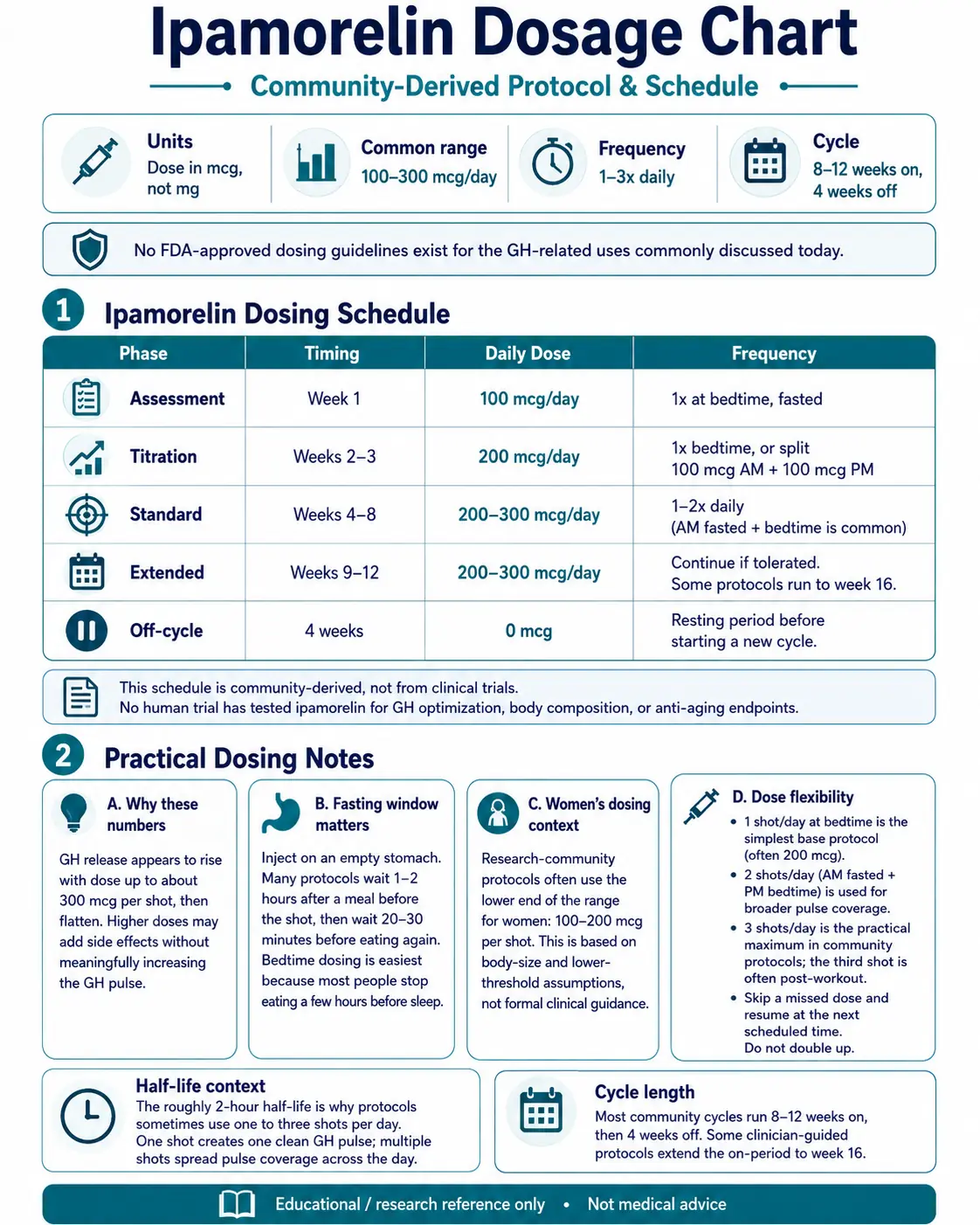
Ipamorelin Dosing Protocol & Schedule
Ipamorelin is dosed in micrograms (mcg), not milligrams. The most common research protocols build slowly from a small starting dose so users can spot side effects early. The schedule below is the structure most often cited in research-community protocols. There are no FDA-approved dosing guidelines for the GH-related uses people care about today.
Ipamorelin Dosing Schedule (Community-Derived)
Phase
Assessment
Timing
Week 1
Daily Dose
100 mcg/day
Frequency
1x at bedtime, fasted
Phase
Titration
Timing
Weeks 2-3
Daily Dose
200 mcg/day
Frequency
1x bedtime, or split 100 mcg AM + 100 mcg PM
Phase
Standard
Timing
Weeks 4-8
Daily Dose
200-300 mcg/day
Frequency
1-2x daily (AM fasted + bedtime is common)
Phase
Extended
Timing
Weeks 9-12
Daily Dose
200-300 mcg/day
Frequency
Continue if tolerated. Some protocols run to week 16.
Phase
Off-cycle
Timing
4 weeks
Daily Dose
0 mcg
Frequency
Resting period before starting a new cycle.
| Phase | Timing | Daily Dose | Frequency |
|---|---|---|---|
| Assessment | Week 1 | 100 mcg/day | 1x at bedtime, fasted |
| Titration | Weeks 2-3 | 200 mcg/day | 1x bedtime, or split 100 mcg AM + 100 mcg PM |
| Standard | Weeks 4-8 | 200-300 mcg/day | 1-2x daily (AM fasted + bedtime is common) |
| Extended | Weeks 9-12 | 200-300 mcg/day | Continue if tolerated. Some protocols run to week 16. |
| Off-cycle | 4 weeks | 0 mcg | Resting period before starting a new cycle. |
This schedule is community-derived, not from clinical trials. No human trial has tested ipamorelin for GH optimization, body composition, or anti-aging endpoints.
Why these numbers
Research suggests GH release climbs with dose up to about 300 mcg per shot, then flattens out. Going higher does not add more GH pulse, but it can add more side effects. That is the practical ceiling in community protocols.
The two-hour half-life is the reason for the one-to-three-shots-per-day range. One shot gives one clean GH pulse. Two or three shots stretch the GH-pulse coverage across the day. Many users start with the bedtime shot only because GH naturally surges during deep sleep.
The fasting window matters
Inject on an empty stomach. Most protocols wait 1-2 hours after a meal before the shot, then wait 20-30 minutes before eating again. Food raises insulin, and insulin blunts the GH pulse. The bedtime shot is easiest to time because most people stop eating a few hours before sleep.
Women's dosing context
Research-community protocols often use the lower end of the dose range for women: 100-200 mcg per shot. The reason cited is body size and a generally lower baseline GH-response threshold. This is not a formal guideline. No clinical trial has set a sex-specific dose for ipamorelin.
Dose-flexibility notes
- One shot per day at bedtime is the simplest base protocol (often 200 mcg).
- Two shots per day (AM fasted + PM bedtime) is used when protocols target sustained pulse coverage.
- Three shots per day is the maximum in community protocols. The third shot is often timed post-workout.
- Skip a missed dose and resume at the next scheduled time. Do not double up.
Cycle length
Most community cycles run 8 to 12 weeks on, then 4 weeks off. The off-cycle is intended to keep the GH receptor responsive. Some clinician-guided protocols extend the on-period to week 16.
Ipamorelin Reconstitution Guide
Ipamorelin ships as a freeze-dried powder. You add bacteriostatic (BAC) water to dissolve it before drawing your shot. The table below lists common vial sizes, the BAC water volume that gives clean math, the resulting concentration, and the syringe units on a U-100 insulin syringe.
Ipamorelin Reconstitution — Vial Size & Syringe Units
Vial Size
2 mg
BAC Water
1.0 mL
Concentration
2,000 mcg/mL
100 mcg
0.05 mL (5 units)
200 mcg
0.10 mL (10 units)
300 mcg
0.15 mL (15 units)
Vial Size
5 mg
BAC Water
2.0 mL
Concentration
2,500 mcg/mL
100 mcg
0.04 mL (4 units)
200 mcg
0.08 mL (8 units)
300 mcg
0.12 mL (12 units)
Vial Size
10 mg
BAC Water
3.0 mL
Concentration
3,333 mcg/mL
100 mcg
0.03 mL (3 units)
200 mcg
0.06 mL (6 units)
300 mcg
0.09 mL (9 units)
| Vial Size | BAC Water | Concentration | 100 mcg | 200 mcg | 300 mcg |
|---|---|---|---|---|---|
| 2 mg | 1.0 mL | 2,000 mcg/mL | 0.05 mL (5 units) | 0.10 mL (10 units) | 0.15 mL (15 units) |
| 5 mg | 2.0 mL | 2,500 mcg/mL | 0.04 mL (4 units) | 0.08 mL (8 units) | 0.12 mL (12 units) |
| 10 mg | 3.0 mL | 3,333 mcg/mL | 0.03 mL (3 units) | 0.06 mL (6 units) | 0.09 mL (9 units) |
Units shown are for a standard U-100 insulin syringe (100 units = 1.0 mL). Round to the nearest whole unit when drawing.
Step-by-step reconstitution
- 01
Bring the vial to room temperature
Let the ipamorelin vial sit out for 5-10 minutes before mixing.
- 02
Swab both stoppers
Wipe the rubber tops of the peptide vial and the BAC water vial with separate alcohol swabs.
- 03
Draw the BAC water
Pull the planned BAC water volume into a sterile syringe (1.0 mL for 2 mg, 2.0 mL for 5 mg, 3.0 mL for 10 mg).
- 04
Add water slowly
Insert the needle into the peptide vial. Let the water drip slowly down the inside glass wall. Do not blast it directly onto the powder.
- 05
Swirl gently
Roll or swirl the vial between your fingers until the powder fully dissolves. Do not shake.
- 06
Check the solution
It should be clear and colorless with no floating particles.
- 07
Label and store
Write the concentration and date on the vial. Refrigerate at 2-8C. Use within about 28 days.
Need custom math?
If your vial size, BAC water volume, or target dose does not match the table, use the Pep Pal calculator for exact syringe units.
Ipamorelin Dosage Chart
This ipamorelin dosage chart summarizes the common research-use dose range from 100 mcg to 300 mcg, with dosing frequency shown by protocol style.
How Ipamorelin Works
Ipamorelin works by pressing a specific 'release GH' button in the brain. That button is a receptor on the pituitary gland called GHS-R1a. It is the same receptor that the hunger hormone ghrelin uses.
When ipamorelin binds to GHS-R1a, the pituitary releases a pulse of growth hormone. The pulse mimics the way your body already releases GH on its own. It does not flood the system with a constant supply, which is what direct GH injections do.
The somatostatin brake
Your body has a natural 'brake' on GH release. The brake is a hormone called somatostatin. Ipamorelin appears to ease that brake during a short window. This is one reason bedtime dosing is common — it lines up with a natural dip in somatostatin activity.
Downstream IGF-1
Once GH is released, the liver converts most of it into IGF-1. IGF-1 is the second signal that drives most of the effects people link to GH — muscle protein building, fat metabolism, and tissue repair. These effects stay under the body's normal feedback controls, so they scale with the GH pulse rather than running unchecked.
What it does not do
The defining feature of ipamorelin is what it leaves alone. In lab studies by Raun et al. (1998), ipamorelin did not raise cortisol, ACTH, or prolactin even at doses many times higher than needed to release GH. Other GH-releasing peptides (GHRP-2 and GHRP-6) do raise these hormones. That clean profile is the main reason ipamorelin is often the first-choice GHRP in research protocols.
Targets the GHS-R1a (ghrelin) receptor
Same receptor as ghrelin, located in the pituitary and hypothalamus.
Triggers a pulse, not a flood
GH release is short and natural, peaking within ~30-40 minutes.
Does not raise stress hormones
Cortisol, ACTH, and prolactin stay flat at standard doses.
Short half-life (~2 hours)
Why dosing is built around clean pulses, not steady levels.
Ipamorelin Supplies Needed
These supply estimates are based on 200 mcg per day from a 10 mg vial reconstituted with 3.0 mL BAC water. If you use a 5 mg or 2 mg vial, the BAC water and syringe counts stay the same — only the vial count changes.
Recommended Supply
Use discount code PEPPAL at eligible peptide supplier checkouts.

Ipamorelin 10mg supply

SiPhox Health At-Home Blood Test
Injection Supplies
Disclosure: supply links may earn PDP a commission at no cost to you.
Ipamorelin Vials (10 mg)
A 10 mg vial yields about 50 days of dosing at 200 mcg/day. Add margin for priming loss.
| Cycle length | Planning note |
|---|---|
4 weeks 1 vial | 28 shots needed; 1 vial covers ~50 doses. |
8-12 weeks 2 vials | 8 weeks: 56 shots needed; 2 vials cover ~100 doses.; 12 weeks: 84 shots needed; 2 vials cover ~100 doses with some margin. |
4 weeks
1 vial
28 shots needed; 1 vial covers ~50 doses.
8-12 weeks
2 vials
8 weeks: 56 shots needed; 2 vials cover ~100 doses.; 12 weeks: 84 shots needed; 2 vials cover ~100 doses with some margin.
Insulin Syringes (U-100, 0.3 mL / 30-unit preferred)
One syringe per shot. Use 30-unit insulin syringes for small draws.
| Cycle length | Planning note |
|---|---|
4 weeks 28 syringes | 1 shot per day. |
8 weeks 56 syringes | 1 shot per day. |
12 weeks 84 syringes | 1 shot per day; recommend 1 x 100-count box. |
4 weeks
28 syringes
1 shot per day.
8 weeks
56 syringes
1 shot per day.
12 weeks
84 syringes
1 shot per day; recommend 1 x 100-count box.
Bacteriostatic Water (10 mL bottles)
Use 3.0 mL per 10 mg vial. One 10 mL bottle covers about 3 vial reconstitutions.
| Cycle length | Planning note |
|---|---|
4-12 weeks 1 x 10 mL bottle | 4 weeks: 1 vial uses 3 mL.; 8 weeks: 2 vials use 6 mL total.; 12 weeks: 2 vials use 6 mL; one bottle gives margin for losses. |
4-12 weeks
1 x 10 mL bottle
4 weeks: 1 vial uses 3 mL.; 8 weeks: 2 vials use 6 mL total.; 12 weeks: 2 vials use 6 mL; one bottle gives margin for losses.
Round up for priming losses, dropped syringes, and protocol adjustments.
Who Ipamorelin Is For and Who Should Avoid It
Ipamorelin is a research compound. It is not approved for any human use. Even within research contexts, there are clear groups for whom the risk-benefit math does not work.
Avoid use in
- Active or recent cancer. GH and IGF-1 can support cell growth. Anyone with current or recent cancer should avoid GH-raising compounds.
- Uncontrolled blood sugar disease (type 1 or type 2 diabetes). GH affects insulin sensitivity. Glucose control can shift.
- Severe heart disease. GH can cause fluid retention. That can stress an already weak heart.
- Pregnancy or breastfeeding. There is no safety data for ipamorelin in either group.
- Children or teens. GH and IGF-1 directly affect growth plates. Ipamorelin is not for anyone still growing.
- Competitive athletes. Ipamorelin and all ghrelin mimetics are banned under WADA rules.
Use extra caution if
- You have a strong family history of cancer.
- You are pre-diabetic or have a fasting glucose near the high end of normal.
- You take medications that affect blood sugar (insulin, GLP-1s, steroids).
- You have sleep apnea — GH can worsen it.
- You have carpal tunnel symptoms — GH-related fluid retention can flare them.
Monitoring boundaries
Some practitioners review fasting glucose and HbA1c before and during extended ipamorelin protocols. This page is not medical advice. Any decision to start, continue, or stop a research compound is between an individual and their licensed clinician.
Ipamorelin Side Effects & Safety
Ipamorelin's side-effect profile is cleaner than most GH-releasing peptides. Most reports describe mild, short-lived effects. Side effects still happen, especially at higher doses or in the first weeks of use.
Commonly reported (mild)
- Mild headache, usually short-lived.
- Light water retention or bloating.
- Mild appetite increase. Ipamorelin is less appetite-driving than GHRP-6.
- Tingling in hands or feet, mostly at the start of a cycle.
Reported with higher doses or longer use
- Mild joint stiffness.
- Temporary fatigue.
- Carpal tunnel-style symptoms (linked to GH-related fluid retention).
Injection-site reactions
Some users report mild redness, stinging, or a small bump at the injection site. Rotating sites and keeping sterile technique reduces how often this happens.
What ipamorelin does not raise
Unlike GHRP-2 and GHRP-6, ipamorelin did not raise cortisol (a stress hormone), ACTH, or prolactin in lab testing at standard GH-releasing doses (Raun et al. 1998). That cleaner profile is the main reason it is generally better tolerated than the other GHRPs.
Clinical tolerability data
In the Beck et al. 2014 Phase II trial (117 bowel surgery patients, NCT00672074), ipamorelin given as 0.03 mg/kg IV twice daily for up to 7 days produced treatment-related adverse event rates similar to placebo. The trial did not meet its efficacy endpoint, but tolerability was not the reason the program was discontinued.
Quality-control risk
Most ipamorelin sold today comes from research-use suppliers, not pharmacies. Quality varies. Verify a recent third-party Certificate of Analysis (COA) before sourcing. For broader safety context, see the PepPal peptide side effects guide.
Ipamorelin Timeline & What to Monitor
Clinical trials never tracked body-composition or recovery endpoints for ipamorelin, so there is no validated 'timeline' for those uses. The timeline below combines published pharmacokinetic data with research-community reports.
Ipamorelin Response Timeline
Phase
Single shot
Window
30-40 minutes
What People Report
GH peaks. No noticeable feeling at standard doses. Some report a slight 'pulse' sensation or fullness.
Phase
Day 1-7
Window
Early signal
What People Report
Mild headache or tingling possible. Some report deeper sleep on bedtime dosing.
Phase
Week 2-4
Window
Body acclimates
What People Report
Most early side effects fade. Sleep changes (if present) tend to stabilize.
Phase
Week 4-8
Window
Reported changes
What People Report
Some users report changes in recovery, sleep quality, or skin. These reports are variable and not from controlled studies.
Phase
Week 8-12
Window
Standard cycle end
What People Report
Most community protocols stop here for a 4-week off-cycle.
| Phase | Window | What People Report |
|---|---|---|
| Single shot | 30-40 minutes | GH peaks. No noticeable feeling at standard doses. Some report a slight 'pulse' sensation or fullness. |
| Day 1-7 | Early signal | Mild headache or tingling possible. Some report deeper sleep on bedtime dosing. |
| Week 2-4 | Body acclimates | Most early side effects fade. Sleep changes (if present) tend to stabilize. |
| Week 4-8 | Reported changes | Some users report changes in recovery, sleep quality, or skin. These reports are variable and not from controlled studies. |
| Week 8-12 | Standard cycle end | Most community protocols stop here for a 4-week off-cycle. |
No clinical trial has validated body-composition, recovery, or anti-aging endpoints for ipamorelin. Community reports are not the same as proof.
Markers some practitioners review
- Fasting glucose and HbA1c — GH affects insulin sensitivity.
- IGF-1 — direct downstream marker of GH activity. Baseline and follow-up readings give a measurable signal.
- Body composition (DEXA or similar) — only useful with a long enough window to detect change.
Stop or pause the protocol if persistent headaches, glucose spikes, vision changes, or carpal tunnel symptoms appear. This guide is research context, not a treatment plan.
Ipamorelin Clinical Evidence Context
Ipamorelin's published research is narrow. Early studies showed that it works as a selective GH secretagogue and that it has a short, well-characterized half-life. The clinical program then tested it for postoperative ileus — a digestive condition where the gut is slow to restart after surgery. The program did not show efficacy and was discontinued. No clinical trial has tested ipamorelin for the GH-optimization, body-composition, or anti-aging uses people care about today.
Key Ipamorelin Clinical Research
Study
Raun et al. 1998 (Eur J Endocrinol)
Type
Preclinical characterization
Subjects
Rats and swine
Key Finding
Defined ipamorelin as a selective GH secretagogue. No ACTH or cortisol rise even at very high multiples of the GH-effective dose.
Study
Gobburu et al. 1999 (Pharm Res)
Type
Phase I PK/PD
Subjects
40 healthy male volunteers
Key Finding
Dose-proportional pharmacokinetics. Terminal half-life ~2 hours. Peak GH near 40 minutes post-shot.
Study
Beck et al. 2014 (Int J Colorectal Dis)
Type
Phase II
Subjects
117 bowel surgery patients (NCT00672074)
Key Finding
Ipamorelin 0.03 mg/kg IV twice daily was tolerated similarly to placebo. Did not show efficacy for postoperative ileus.
Study
ClinicalTrials.gov NCT01280344
Type
Phase II
Subjects
Postoperative ileus
Key Finding
Program discontinued after lack of efficacy signal for the POI endpoint.
| Study | Type | Subjects | Key Finding |
|---|---|---|---|
| Raun et al. 1998 (Eur J Endocrinol) | Preclinical characterization | Rats and swine | Defined ipamorelin as a selective GH secretagogue. No ACTH or cortisol rise even at very high multiples of the GH-effective dose. |
| Gobburu et al. 1999 (Pharm Res) | Phase I PK/PD | 40 healthy male volunteers | Dose-proportional pharmacokinetics. Terminal half-life ~2 hours. Peak GH near 40 minutes post-shot. |
| Beck et al. 2014 (Int J Colorectal Dis) | Phase II | 117 bowel surgery patients (NCT00672074) | Ipamorelin 0.03 mg/kg IV twice daily was tolerated similarly to placebo. Did not show efficacy for postoperative ileus. |
| ClinicalTrials.gov NCT01280344 | Phase II | Postoperative ileus | Program discontinued after lack of efficacy signal for the POI endpoint. |
Source links are in the Sources & Research section.
What this means in plain English
Ipamorelin clearly works as a GH-releasing peptide. The pharmacology is solid. But the only Phase II trial tested it for a use most people are not interested in (gut recovery after surgery), and that trial did not work. The dose ranges, schedules, and cycle lengths in this guide come from community protocols, not from validated clinical evidence.
Ipamorelin Storage & Handling
Ipamorelin Storage Guidelines
State
Lyophilized powder
Temperature
-4F (-20C)
Duration
Long-term (months to years)
State
Lyophilized powder
Temperature
36-46F (2-8C)
Duration
Months
State
Lyophilized powder
Temperature
Room temperature
Duration
Weeks (shipping window)
State
Reconstituted (liquid)
Temperature
36-46F (2-8C)
Duration
Up to 28 days
State
Reconstituted, frozen aliquots
Temperature
-4F (-20C)
Duration
3-4 months
| State | Temperature | Duration |
|---|---|---|
| Lyophilized powder | -4F (-20C) | Long-term (months to years) |
| Lyophilized powder | 36-46F (2-8C) | Months |
| Lyophilized powder | Room temperature | Weeks (shipping window) |
| Reconstituted (liquid) | 36-46F (2-8C) | Up to 28 days |
| Reconstituted, frozen aliquots | -4F (-20C) | 3-4 months |
Protect from light. Avoid repeated freeze-thaw cycles. Discard any solution that is cloudy, discolored, or has visible particles.
Keep the vial upright in the back of the refrigerator, not in the door. Use bacteriostatic water (not sterile water) for multi-dose handling — the benzyl alcohol preservative helps with shelf life.
Ipamorelin Protocol Mistakes & Troubleshooting
Most ipamorelin protocol problems trace back to one of five issues. Each is fixable.
1. Eating too close to the shot
If insulin spikes from a meal are still active, the GH pulse gets blunted. Wait at least 1-2 hours after a meal before the shot. Wait 20-30 minutes after the shot before eating.
2. Cloudy or discolored solution
Reconstituted ipamorelin should be clear and colorless. If the solution is cloudy, has particles, or has changed color, do not use it. Reconstitute a fresh vial.
3. Wrong BAC water volume
Adding more BAC water does not make the peptide weaker — it changes the concentration, so the syringe units for a given dose shift. Re-check the math against the reconstitution table. The Pep Pal calculator covers custom volumes.
4. Missed shots
Skip the missed shot and resume the next scheduled time. Do not double up. A missed shot does not break the cycle.
5. Storage mistakes
Reconstituted ipamorelin needs to stay refrigerated (36-46F / 2-8C). Leaving it on the counter shortens its life. Freezing the reconstituted vial can damage the peptide unless you freeze it in single-use aliquots.
Headaches won't stop?
Drop back to the previous dose for a few days. Most headaches fade within the first 1-2 weeks. Persistent headaches at low doses are a reason to pause and reassess.
Ipamorelin Regulatory Status
Ipamorelin is not FDA-approved for any human use. Its status under FDA pharmacy compounding rules has been actively debated, and the rules have shifted more than once. This section is current as of May 2026.
The 503A compounding timeline
- September 2023 — FDA placed ipamorelin (and several other peptides) on Category 2 of the interim 503A bulks list. Category 2 means 'do not compound.'
- September 27, 2024 — Ipamorelin was removed from Category 2 after the original nominator withdrew the nomination. AOD-9604, CJC-1295, thymosin alpha-1, and Selank were removed at the same time.
- October 29, 2024 — Pharmacy Compounding Advisory Committee (PCAC) reviewed ipamorelin for inclusion in the 503A bulks regulation. FDA recommended against inclusion.
- February 27, 2026 — HHS Secretary publicly stated intent to broaden peptide access. The FDA has not formally added ipamorelin to Category 1 as of May 2026.
What this means in practice: ipamorelin is currently not eligible to be compounded by 503A pharmacies, but it is also no longer on the 'do not compound' Category 2 list. Most ipamorelin in circulation is sold by research-use suppliers, not pharmacies.
Anti-doping status
Ipamorelin and all ghrelin mimetics are prohibited substances under WADA rules, both in-competition and out-of-competition. Athletes subject to drug testing should not use ipamorelin.
International status
Ipamorelin is not approved as a medicine in the EU, UK, Canada, or Australia. Importation rules vary. This is a research compound everywhere.
Ipamorelin vs CJC-1295 vs Tesamorelin vs GHRP-2 vs GHRP-6
Ipamorelin is one of several compounds that raise growth hormone. Each works a bit differently. The right comparison depends on what the protocol is built around.
Ipamorelin vs GHRP-2 and GHRP-6
GHRP Family — Side-by-Side
Feature
Receptor
Ipamorelin
GHS-R1a
GHRP-2
GHS-R1a
GHRP-6
GHS-R1a
Feature
Half-life
Ipamorelin
~2 hours
GHRP-2
~25-30 minutes
GHRP-6
~20-30 minutes
Feature
Dosing
Ipamorelin
1-3x daily (SubQ)
GHRP-2
2-3x daily (SubQ)
GHRP-6
2-3x daily (SubQ)
Feature
Standard dose
Ipamorelin
100-300 mcg
GHRP-2
100-300 mcg
GHRP-6
100-300 mcg
Feature
Cortisol effect
Ipamorelin
None at standard doses
GHRP-2
Yes
GHRP-6
Yes
Feature
Prolactin effect
Ipamorelin
None
GHRP-2
Moderate
GHRP-6
Minimal
Feature
Appetite effect
Ipamorelin
Mild
GHRP-2
Moderate
GHRP-6
Strong
Feature
FDA status
Ipamorelin
Not approved
GHRP-2
Not approved
GHRP-6
Not approved
| Feature | Ipamorelin | GHRP-2 | GHRP-6 |
|---|---|---|---|
| Receptor | GHS-R1a | GHS-R1a | GHS-R1a |
| Half-life | ~2 hours | ~25-30 minutes | ~20-30 minutes |
| Dosing | 1-3x daily (SubQ) | 2-3x daily (SubQ) | 2-3x daily (SubQ) |
| Standard dose | 100-300 mcg | 100-300 mcg | 100-300 mcg |
| Cortisol effect | None at standard doses | Yes | Yes |
| Prolactin effect | None | Moderate | Minimal |
| Appetite effect | Mild | Moderate | Strong |
| FDA status | Not approved | Not approved | Not approved |
All three target the same receptor. Ipamorelin's value is the clean hormonal profile.
Ipamorelin vs CJC-1295
Ipamorelin and CJC-1295 are not interchangeable — they work on different receptors. Ipamorelin presses the GHS-R1a (ghrelin) receptor. CJC-1295 presses the GHRH receptor. Most protocols stack them together because pressing both receptors at once creates a larger GH pulse than either one alone.
A common stack uses 100 mcg CJC-1295 (no DAC, also called Mod GRF 1-29) plus 200 mcg ipamorelin in the same shot at bedtime. See the dedicated CJC-1295 + Ipamorelin stack guide for the full protocol breakdown.
Ipamorelin vs Tesamorelin and Sermorelin
Tesamorelin and sermorelin are both GHRH analogs — they work on the GHRH receptor, not the GHS-R1a receptor. Tesamorelin is the only one of this group with an FDA approval (for HIV-associated lipodystrophy). Ipamorelin is not interchangeable with either. Different receptor, different protocol, different evidence base.
If you are comparing a GHRH analog plus a ghrelin-mimetic pairing, see the tesamorelin + ipamorelin blend protocol for ratio-specific reconstitution math and cycle planning.
See the compound-specific guides for Tesamorelin, Sermorelin, CJC-1295 No DAC, and CJC-1295 DAC.
Ipamorelin Blood Tests & Monitoring
Ipamorelin is usually discussed as a GH secretagogue pathway peptide. Monitoring focuses on IGF-1, glucose handling, lipids, thyroid context, appetite, and fluid-retention symptoms.
Blood test markers to discuss with a clinician
Marker
IGF-1
Why it matters
Shows the downstream signal after repeated GH secretagogue activity.
Timing
Follow-up
Marker
A1c
Why it matters
Tracks longer-term glucose control because GH-axis changes may affect insulin sensitivity.
Timing
Baseline
Marker
Fasting glucose
Why it matters
Gives a current glucose snapshot during GH-axis protocols.
Timing
Follow-up
Marker
Lipid panel
Why it matters
Reviews cholesterol and triglyceride trends during body-composition changes.
Timing
Baseline
Marker
TSH and free T4
Why it matters
Helps separate thyroid-related symptoms from GH-axis effects.
Timing
Optional
| Marker | Why it matters | Timing |
|---|---|---|
| IGF-1 | Shows the downstream signal after repeated GH secretagogue activity. | Follow-up |
| A1c | Tracks longer-term glucose control because GH-axis changes may affect insulin sensitivity. | Baseline |
| Fasting glucose | Gives a current glucose snapshot during GH-axis protocols. | Follow-up |
| Lipid panel | Reviews cholesterol and triglyceride trends during body-composition changes. | Baseline |
| TSH and free T4 | Helps separate thyroid-related symptoms from GH-axis effects. | Optional |
Monitoring guidance is GH-secretagogue pathway-based because ipamorelin-specific clinical monitoring standards are limited.
At-home blood test option
Easy at home option to monitor core metrics during research cycles.

Partner link: PDP may earn a commission at no cost to you.
Simple timing framework
Baseline
Discuss baseline labs before starting, especially with diabetes risk, sleep apnea, edema, cancer history, or thyroid disease.
Follow-up
Re-check IGF-1 and metabolic markers after 6-8 weeks or after meaningful dose changes.
Longer term
For longer protocols, review trends every 3-6 months with a clinician.
How to interpret the labs
- IGF-1 trends should be interpreted with age, symptoms, and baseline levels.
- Appetite, water retention, sleep apnea symptoms, and glucose changes are important pathway signals.
- Cancer history or active malignancy concerns need clinician review before GH-axis stimulation is considered.
Do not wait for routine labs
Rapid swelling, severe headaches, vision changes, chest pain, or shortness of breath needs medical review. New numbness, wrist pain, or worsening sleep apnea symptoms should be discussed with a clinician.
FAQ
Q1: What is ipamorelin?
Ipamorelin is a research peptide that signals the pituitary to release the body's own growth hormone in natural pulses. It is also called NNC 26-0161. It is the most selective member of the GHRP family — it raises GH without raising cortisol, prolactin, or other stress hormones.
Q2: What is the starting dose of ipamorelin?
The common starting dose in community protocols is 100 mcg per shot, once daily at bedtime, on an empty stomach, for about one week. Most protocols then step up to 200 mcg per shot. For custom vial math, use the Pep Pal calculator.
Q3: What is the ipamorelin dosage chart by vial size?
For a 2 mg vial with 1.0 mL BAC water: 200 mcg = 10 units. For a 5 mg vial with 2.0 mL BAC water: 200 mcg = 8 units. For a 10 mg vial with 3.0 mL BAC water: 200 mcg = 6 units. Units are based on a standard U-100 insulin syringe. See the full reconstitution table on this page.
Q4: How is ipamorelin reconstituted?
Common setups are 1.0 mL BAC water for a 2 mg vial, 2.0 mL BAC water for a 5 mg vial, and 3.0 mL BAC water for a 10 mg vial. Drip the BAC water down the inside wall of the vial. Swirl gently — do not shake. Refrigerate and use within about 28 days.
Q5: What is ipamorelin's half-life?
Ipamorelin has a terminal plasma half-life of about 2 hours in human pharmacokinetic data (Gobburu et al. 1999). GH peaks around 30-40 minutes after the shot and returns to baseline within 2-3 hours.
Q6: Is ipamorelin FDA-approved?
No. Ipamorelin is not FDA-approved for any human use. It reached Phase II clinical trials for postoperative ileus (a gut-recovery condition) and was discontinued after the trial did not show efficacy. Ipamorelin was on the FDA's Category 2 503A compounding list from September 2023 to September 2024, then removed after the nominator withdrew. As of May 2026 it is not Category 1 and is not eligible for 503A compounding. It is also banned under WADA anti-doping rules.
Q7: What is the ipamorelin dosage for women?
Research-community protocols often use 100-200 mcg per shot for women, with the bedtime shot prioritized. The lower end of the dose range is cited for body-size and baseline GH-response reasons. No clinical trial has set a sex-specific dose for ipamorelin. This is not a personal medical recommendation.
Q8: What is the ipamorelin dosing schedule for beginners?
A common beginner schedule is 100 mcg per shot, one shot per day at bedtime, on an empty stomach, for one week. Most protocols then move to 200 mcg per shot in week 2. By week 4, the most common range is 200-300 mcg per shot, one or two times daily. Cycles run 8-12 weeks on, 4 weeks off.
Q9: How is the CJC-1295 + ipamorelin stack dosed?
A frequently cited stack uses 100 mcg CJC-1295 (no DAC, also called Mod GRF 1-29) plus 200 mcg ipamorelin together in a single subcutaneous shot at bedtime. The two peptides press different receptors, so the GH pulse is larger than either one alone. See the CJC-1295 + Ipamorelin stack guide for the full protocol.
Q10: How long is a typical ipamorelin cycle?
Most community protocols run 8 to 12 weeks on, then 4 weeks off. Some clinician-guided protocols extend the on-period to week 16. The off-period is intended to keep the GH receptor responsive.
Q11: What is the maximum daily dose of ipamorelin?
Most research protocols cap single shots around 300 mcg because GH release flattens out beyond that point — more dose does not equal more GH pulse. Daily totals up to 900 mcg (300 mcg three times per day) have been used in community protocols, but tolerability and side-effect risk climb at the high end.
Q12: What are the most common ipamorelin side effects?
The most commonly reported effects are mild headache, light water retention, mild appetite increase, and tingling in the hands or feet. Most fade within the first 1-2 weeks. Unlike GHRP-2 and GHRP-6, ipamorelin did not raise cortisol or prolactin in clinical testing at standard doses.
Q13: How does ipamorelin compare to CJC-1295?
Ipamorelin and CJC-1295 are not interchangeable. They work on different receptors. Ipamorelin presses the GHS-R1a (ghrelin) receptor. CJC-1295 presses the GHRH receptor. They are most often used together because pressing both receptors at once produces a larger GH pulse than either one alone.
Q14: How should reconstituted ipamorelin be stored?
Store reconstituted ipamorelin at 36-46F (2-8C), protected from light, and use within about 28 days. Avoid repeated freeze-thaw cycles unless freezing in single-use aliquots.
Q15: Where can I calculate exact syringe units?
Use the Pep Pal reconstitution calculator to convert vial size, BAC water volume, and target dose into exact syringe units.
Sources & Research
- 1. Raun K, Hansen BS, Johansen NL, et al. Ipamorelin, the first selective growth hormone secretagogue. European Journal of Endocrinology (1998)
- 2. Gobburu JV, Agerso H, Jusko WJ, Ynddal L. Pharmacokinetic-pharmacodynamic modeling of ipamorelin, a growth hormone releasing peptide, in human volunteers. Pharmaceutical Research (1999)
- 3. Beck DE, Sweeney WB, McCarter MD; Ipamorelin 201 Study Group. Prospective, randomized, controlled, proof-of-concept study of the Ghrelin mimetic ipamorelin for the management of postoperative ileus in bowel resection patients. International Journal of Colorectal Disease (2014)
- 4. Ishida J, Saitoh M, Ebner N, et al. Growth hormone secretagogues: history, mechanism of action, and clinical development. JCSM Rapid Communications (2020)
- 5. Hansen BS, Raun K, Nielsen KK, et al. Pharmacological characterisation of a new oral GH secretagogue, NN703. European Journal of Endocrinology (1999)
- 6. ClinicalTrials.gov Safety and Efficacy of Ipamorelin Compared to Placebo for the Recovery of Gastrointestinal Function (NCT01280344). ClinicalTrials.gov (2011)
- 7. ClinicalTrials.gov Ipamorelin in Postoperative Ileus (NCT00672074). ClinicalTrials.gov (2008)
- 8. U.S. Food and Drug Administration Interim Policy on Compounding Using Bulk Drug Substances Under Section 503A — Category 2 list and PCAC review updates. FDA.gov (2026)
- 9. U.S. Food and Drug Administration FDA Summary Report: Ipamorelin Acetate (PCAC review materials). FDA.gov / Regulations.gov (2024)
- 10. Buscail E, Deraison C. Postoperative ileus: A pharmacological perspective. British Journal of Pharmacology (2022)
- 11. Sigalos JT, Pastuszak AW, Khera M. A review of the effects of growth hormone secretagogues on body composition and bone health. Sexual Medicine Reviews (2018)
- 12. World Anti-Doping Agency Prohibited List — S2 Peptide Hormones, Growth Factors, Related Substances and Mimetics (covers ghrelin mimetics). WADA (2026)
Related Dosing Protocols
Educational use only
This guide is an educational research reference, not medical advice or a treatment plan. Ipamorelin is not FDA-approved for any human use.
Calculate ipamorelin vial math
Use the calculator for custom vial size, BAC water volume, and syringe-unit math.
Open CalculatorWritten by Garret Grant
Founder & Lead Researcher · B.S. Civil Engineering, UCLA
Last updated: May 2026
Human-researched and AI-assisted with full editorial review. I verify sources, protocol interpretation, and final judgments personally. See methodology.
Share this page
