Semaglutide Quick Start
Semaglutide is one of the most widely studied weight-loss and diabetes drugs in the world. You may know it as Ozempic for type 2 diabetes, Wegovy for weight management, or Rybelsus for the daily oral tablet. All three are the same active drug. Novo Nordisk makes all of them.
Semaglutide is a GLP-1 receptor agonist. That means it copies a natural gut hormone (GLP-1) that helps the body manage hunger and blood sugar. The injectable version is taken once a week. The Rybelsus and Wegovy pill versions are taken once a day.
Two routes
Injectable (once weekly) and oral tablet (once daily). The two routes use different dose schedules.
Slow start
Doses start very low and step up every 4 weeks. This helps the stomach get used to the drug.
Easy measuring
Most research-use vials work cleanly at 2.5 mg/mL. At that mix, 0.25 mg is 10 units on a U-100 syringe.
Long half-life
About 7 days. Steady levels take roughly 4-5 weeks at each dose.
Status
FDA-approved. Research-use vials from supplier sources are for research only.
Educational reference only
This page is an educational research guide. It is not medical advice and is not a treatment plan. Talk to a qualified clinician before starting any peptide.
Semaglutide Dosing Protocol & Schedule
Semaglutide has two main dosing schedules. The injectable schedule (Wegovy and Ozempic) escalates over about 16 weeks. The oral schedule (Rybelsus for diabetes and the Wegovy 25 mg pill for obesity) escalates daily. Choose the tab below that matches the route you are researching.
Semaglutide Dosing Schedules by Route
Pick the format you are researching. Each tab shows the matching titration schedule.
Subcutaneous injection, once per week. Most common research-use format.
Injectable Titration Schedule
The injectable schedule below is the FDA-approved Wegovy escalation. It runs over about 16 weeks before reaching the 2.4 mg weight-management maintenance dose. Each phase lasts 4 weeks.
Wegovy weight-management titration (2.4 mg target)
Phase
Phase 1 - Initiation
Weeks
Weeks 1-4
Weekly dose
0.25 mg
What to expect
Starting dose for tolerability only. Not a therapeutic dose. Minimal metabolic effect expected.
Phase
Phase 2 - Early escalation
Weeks
Weeks 5-8
Weekly dose
0.5 mg
What to expect
First meaningful dose. Stomach side effects (nausea) most likely to show up here.
Phase
Phase 3 - Mid escalation
Weeks
Weeks 9-12
Weekly dose
1.0 mg
What to expect
Therapeutic range for type 2 diabetes (Ozempic). Appetite suppression often becomes noticeable.
Phase
Phase 4 - High escalation
Weeks
Weeks 13-16
Weekly dose
1.7 mg
What to expect
Approaching the weight-management dose. Weight loss often clearly underway.
Phase
Phase 5 - Maintenance
Weeks
Week 17+
Weekly dose
2.4 mg
What to expect
FDA-approved maintenance dose for weight management (Wegovy). 14.9% mean weight loss at 68 weeks in STEP 1.
| Phase | Weeks | Weekly dose | What to expect |
|---|---|---|---|
| Phase 1 - Initiation | Weeks 1-4 | 0.25 mg | Starting dose for tolerability only. Not a therapeutic dose. Minimal metabolic effect expected. |
| Phase 2 - Early escalation | Weeks 5-8 | 0.5 mg | First meaningful dose. Stomach side effects (nausea) most likely to show up here. |
| Phase 3 - Mid escalation | Weeks 9-12 | 1.0 mg | Therapeutic range for type 2 diabetes (Ozempic). Appetite suppression often becomes noticeable. |
| Phase 4 - High escalation | Weeks 13-16 | 1.7 mg | Approaching the weight-management dose. Weight loss often clearly underway. |
| Phase 5 - Maintenance | Week 17+ | 2.4 mg | FDA-approved maintenance dose for weight management (Wegovy). 14.9% mean weight loss at 68 weeks in STEP 1. |
Type 2 diabetes (Ozempic) uses a similar schedule but caps at 2.0 mg weekly, not 2.4 mg.
Titration Notes
- Titration pacing matters. Slow dose increases reduce stomach side effects. In the STEP trials, side effects were most common during escalation and usually settled by week 20.
- Dose flexibility. FDA labeling allows delaying the dose increase for an extra 4 weeks at the current level if tolerability is an issue. If 2.4 mg is not tolerated, the dose may be temporarily reduced to 1.7 mg for 4 weeks before trying again.
- Missed dose guidance. If a dose is missed and the next scheduled dose is more than 2 days away, the missed dose can be taken as soon as possible. If fewer than 2 days remain, skip the missed dose and stay on the weekly schedule.
- T2D dosing differs. For type 2 diabetes (Ozempic), the maintenance options are 0.5 mg, 1.0 mg, or 2.0 mg weekly. The maximum is 2.0 mg, not 2.4 mg.
Calculate your draw
Use the Pep Pal calculator to convert any vial size, water volume, and target dose into exact syringe units.
Once-daily tablet. Includes Rybelsus (T2D) and the Wegovy 25 mg pill (obesity).
Oral Tablet Titration
Oral semaglutide comes in two FDA-approved forms. Rybelsus is for type 2 diabetes and tops out at 14 mg daily. The Wegovy 25 mg pill was approved in December 2025 for chronic weight management and tops out at 25 mg daily. Both tablets must be taken on an empty stomach with a small sip of water (about 4 oz). Wait 30 minutes before eating, drinking, or taking other medicines.
Wegovy 25 mg pill titration (obesity)
Phase
Phase 1
Weeks
Weeks 1-4
Daily dose
1.5 mg
Notes
Starting dose. Tolerability only.
Phase
Phase 2
Weeks
Weeks 5-8
Daily dose
4 mg
Notes
First step up.
Phase
Phase 3
Weeks
Weeks 9-12
Daily dose
9 mg
Notes
Mid-range dose.
Phase
Phase 4
Weeks
Week 13+
Daily dose
25 mg
Notes
Maintenance dose. 16.6% weight loss at 64 weeks in OASIS 4.
| Phase | Weeks | Daily dose | Notes |
|---|---|---|---|
| Phase 1 | Weeks 1-4 | 1.5 mg | Starting dose. Tolerability only. |
| Phase 2 | Weeks 5-8 | 4 mg | First step up. |
| Phase 3 | Weeks 9-12 | 9 mg | Mid-range dose. |
| Phase 4 | Week 13+ | 25 mg | Maintenance dose. 16.6% weight loss at 64 weeks in OASIS 4. |
Take on an empty stomach with up to 4 oz of plain water. Wait 30 minutes before food, drink, or other oral medication.
Rybelsus titration (type 2 diabetes)
Phase
Phase 1
Duration
Weeks 1-4
Daily dose
3 mg
Notes
Tolerability dose only. No clinical effect expected.
Phase
Phase 2
Duration
Weeks 5-8
Daily dose
7 mg
Notes
First therapeutic dose. May stay here for ongoing T2D control.
Phase
Phase 3
Duration
Week 9+
Daily dose
14 mg
Notes
Maximum FDA-approved Rybelsus dose.
| Phase | Duration | Daily dose | Notes |
|---|---|---|---|
| Phase 1 | Weeks 1-4 | 3 mg | Tolerability dose only. No clinical effect expected. |
| Phase 2 | Weeks 5-8 | 7 mg | First therapeutic dose. May stay here for ongoing T2D control. |
| Phase 3 | Week 9+ | 14 mg | Maximum FDA-approved Rybelsus dose. |
Maximum Rybelsus dose is 14 mg daily. The 25 mg dose is Wegovy oral (obesity), not Rybelsus.
Oral absorption is fragile
Oral semaglutide bioavailability is very low (about 1%) and depends on strict empty-stomach dosing. Eating or drinking too soon after the tablet sharply reduces absorption.
Cycle Guidelines
Common cycle planning for the injectable schedule
Approach
Standard obesity titration
Duration
About 16 weeks to 2.4 mg
Maintenance phase
Ongoing weekly maintenance
Best for
Wegovy-style obesity research
Approach
Standard T2D titration
Duration
About 8-16 weeks to 1.0 or 2.0 mg
Maintenance phase
Ongoing weekly maintenance
Best for
Ozempic-style diabetes research
Approach
Delayed escalation
Duration
Add 4 weeks at any intolerable step
Maintenance phase
Same target maintenance
Best for
When side effects slow the planned ramp
| Approach | Duration | Maintenance phase | Best for |
|---|---|---|---|
| Standard obesity titration | About 16 weeks to 2.4 mg | Ongoing weekly maintenance | Wegovy-style obesity research |
| Standard T2D titration | About 8-16 weeks to 1.0 or 2.0 mg | Ongoing weekly maintenance | Ozempic-style diabetes research |
| Delayed escalation | Add 4 weeks at any intolerable step | Same target maintenance | When side effects slow the planned ramp |
This is an example research plan, not a personal treatment plan. Cycle length depends on the research goal and tolerability.
Semaglutide Reconstitution Guide
Reconstitution applies to lyophilized (freeze-dried) research-grade semaglutide. Brand pens like Ozempic and Wegovy come pre-filled and skip this step. The table below shows how much bacteriostatic water to add to each common vial size and the matching syringe draws for each dose. Units refers to a standard U-100 insulin syringe, where 100 units equals 1.0 mL.
Three concentration options are shown. The standard 2.5 mg/mL mix uses more water and makes small doses easier to measure accurately. The 5.0 mg/mL mix uses less water and gives smaller injection volumes, but it needs more careful syringe handling. Match the concentration to your syringe comfort level.
2.5 mg/mL mix (standard, easier to measure)
Vial size
2 mg
BAC water
0.8 mL
0.25 mg
0.10 mL (10 u)
0.5 mg
0.20 mL (20 u)
1.0 mg
0.40 mL (40 u)
1.7 mg
0.68 mL (68 u)
2.4 mg
0.96 mL (96 u)
Vial size
3 mg
BAC water
1.2 mL
0.25 mg
0.10 mL (10 u)
0.5 mg
0.20 mL (20 u)
1.0 mg
0.40 mL (40 u)
1.7 mg
0.68 mL (68 u)
2.4 mg
0.96 mL (96 u)
Vial size
5 mg
BAC water
2.0 mL
0.25 mg
0.10 mL (10 u)
0.5 mg
0.20 mL (20 u)
1.0 mg
0.40 mL (40 u)
1.7 mg
0.68 mL (68 u)
2.4 mg
0.96 mL (96 u)
| Vial size | BAC water | 0.25 mg | 0.5 mg | 1.0 mg | 1.7 mg | 2.4 mg |
|---|---|---|---|---|---|---|
| 2 mg | 0.8 mL | 0.10 mL (10 u) | 0.20 mL (20 u) | 0.40 mL (40 u) | 0.68 mL (68 u) | 0.96 mL (96 u) |
| 3 mg | 1.2 mL | 0.10 mL (10 u) | 0.20 mL (20 u) | 0.40 mL (40 u) | 0.68 mL (68 u) | 0.96 mL (96 u) |
| 5 mg | 2.0 mL | 0.10 mL (10 u) | 0.20 mL (20 u) | 0.40 mL (40 u) | 0.68 mL (68 u) | 0.96 mL (96 u) |
u = units on a U-100 insulin syringe.
3.33 mg/mL mix (10 mg vial, max 3.0 mL BAC water)
Vial size
10 mg
BAC water
3.0 mL
0.25 mg
0.075 mL (7.5 u)
0.5 mg
0.15 mL (15 u)
1.0 mg
0.30 mL (30 u)
1.7 mg
0.51 mL (51 u)
2.4 mg
0.72 mL (72 u)
| Vial size | BAC water | 0.25 mg | 0.5 mg | 1.0 mg | 1.7 mg | 2.4 mg |
|---|---|---|---|---|---|---|
| 10 mg | 3.0 mL | 0.075 mL (7.5 u) | 0.15 mL (15 u) | 0.30 mL (30 u) | 0.51 mL (51 u) | 0.72 mL (72 u) |
A 10 mg vial cannot use 4.0 mL BAC water when the vial capacity is 3.0 mL.
5.0 mg/mL mix (concentrated, smaller draws)
Vial size
5 mg
BAC water
1.0 mL
0.25 mg
0.05 mL (5 u)
0.5 mg
0.10 mL (10 u)
1.0 mg
0.20 mL (20 u)
1.7 mg
0.34 mL (34 u)
2.4 mg
0.48 mL (48 u)
Vial size
10 mg
BAC water
2.0 mL
0.25 mg
0.05 mL (5 u)
0.5 mg
0.10 mL (10 u)
1.0 mg
0.20 mL (20 u)
1.7 mg
0.34 mL (34 u)
2.4 mg
0.48 mL (48 u)
| Vial size | BAC water | 0.25 mg | 0.5 mg | 1.0 mg | 1.7 mg | 2.4 mg |
|---|---|---|---|---|---|---|
| 5 mg | 1.0 mL | 0.05 mL (5 u) | 0.10 mL (10 u) | 0.20 mL (20 u) | 0.34 mL (34 u) | 0.48 mL (48 u) |
| 10 mg | 2.0 mL | 0.05 mL (5 u) | 0.10 mL (10 u) | 0.20 mL (20 u) | 0.34 mL (34 u) | 0.48 mL (48 u) |
Concentrated mixes need more precise syringe handling, especially at the 0.25 mg starter dose.
Reconstitution Steps
- 01
Bring to room temperature
Let the lyophilized vial and BAC water sit at room temperature for a few minutes before mixing.
- 02
Clean both stoppers
Wipe the vial stopper and the BAC water stopper with alcohol swabs. Let them air-dry for about 10 seconds.
- 03
Draw the BAC water
Use a sterile syringe to draw the calculated water volume. For a 5 mg vial at 2.5 mg/mL, draw 2.0 mL.
- 04
Inject down the inside wall
Push the water slowly down the inside of the vial wall. Do not spray the powder directly.
- 05
Swirl, do not shake
Gently swirl until the powder fully dissolves. Do not shake. The solution should be clear and colorless with no visible particles.
- 06
Label the vial
Write the peptide name, the concentration (for example, 2.5 mg/mL), and the reconstitution date on the vial.
- 07
Refrigerate
Store at 2-8C (35.6-46.4F) and use within 28 days. Do not freeze reconstituted solution.
Need exact units for a different vial?
Use the free Peptide Reconstitution Calculator. Enter vial size, water volume, and target dose. The calculator returns the exact syringe draw.
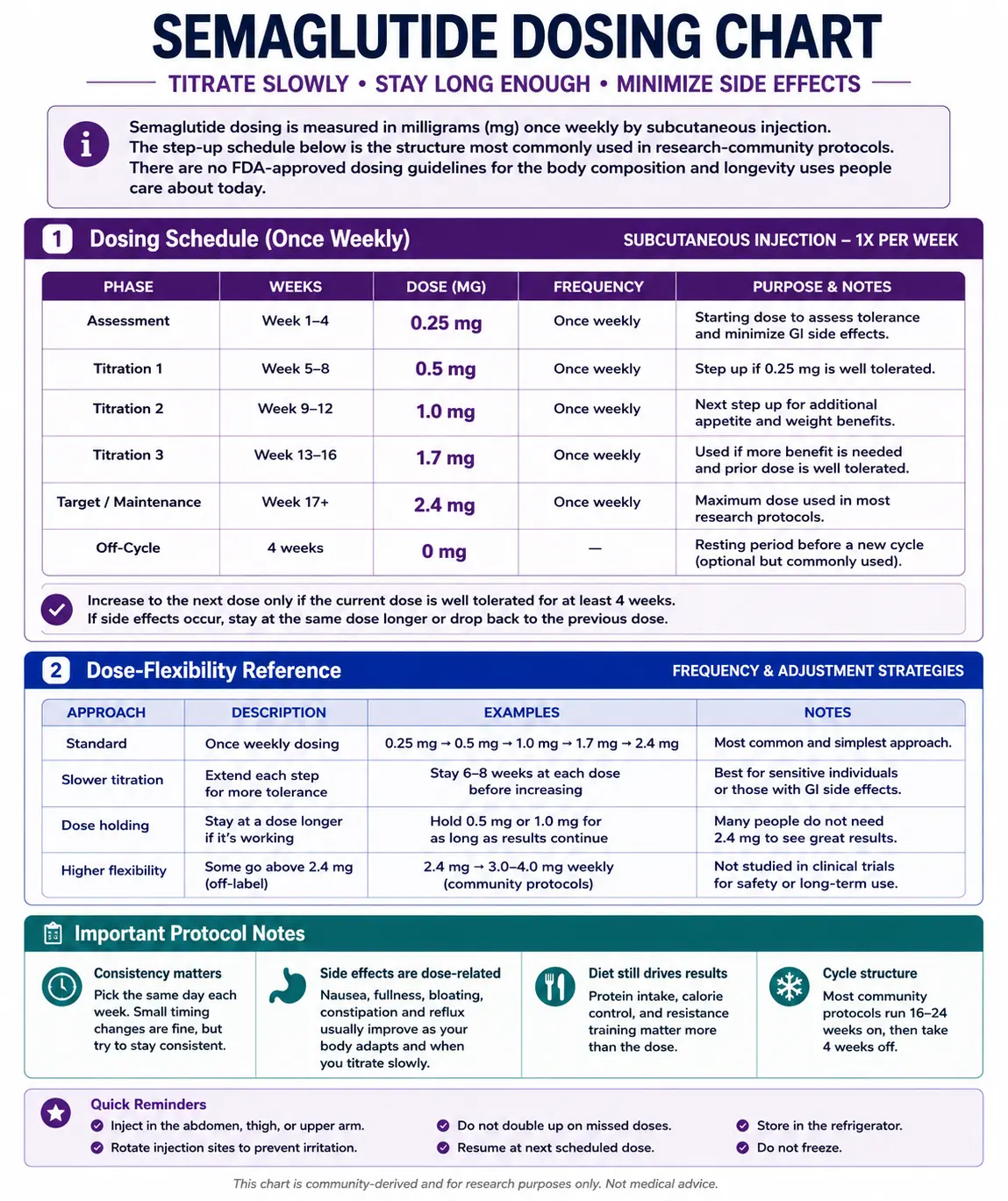
Semaglutide Dosage Chart
This semaglutide dosage chart summarizes the standard once-weekly injectable titration schedule from 0.25 mg up to 2.4 mg, with dose escalation shown by week range.
How Semaglutide Works
Semaglutide turns on a single target called the GLP-1 receptor. That receptor sits on cells in several different organs, which is why semaglutide affects hunger, blood sugar, the stomach, and the heart all at once.
Two small changes to the natural GLP-1 molecule give semaglutide its long shelf life in the body. First, a tweak in the peptide chain blocks the enzyme (DPP-4) that normally breaks down GLP-1 within minutes. Second, a fatty acid chain lets semaglutide bind to albumin, a protein in the blood. That binding slows clearance and stretches the half-life to about 7 days, which is why one weekly injection is enough.
Brain - appetite and fullness
GLP-1 receptors in the hypothalamus and brainstem signal fullness. Semaglutide turning these on reduces hunger and lowers calories eaten. In STEP 1, participants on 2.4 mg lost an average of 14.9% of body weight over 68 weeks.
Pancreas - glucose-dependent insulin
In the pancreas, semaglutide triggers insulin release - but only when blood sugar is already high. This 'glucose-dependent' behavior makes dangerous low blood sugar much less likely than with some older diabetes drugs. In the SUSTAIN trials, semaglutide cut HbA1c by 1.5-1.9% at the 0.5-1.0 mg doses.
Stomach - slower gastric emptying
Semaglutide slows how fast the stomach empties. That stretches fullness after meals and blunts blood sugar spikes. It also explains the nausea and vomiting that are most common during dose escalation and usually fade with time.
Heart - cardiovascular protection
Semaglutide protects the heart and blood vessels in ways that go beyond weight loss alone. The SELECT trial showed a 20% drop in major heart events (heart attack, stroke, cardiovascular death) in people with obesity and existing heart disease. The benefit appeared even in people who did not lose much weight.
Semaglutide Supplies Needed
Plan supplies around the injectable schedule above: weekly subcutaneous injections from a reconstituted lyophilized vial. The math below assumes 2.5 mg/mL concentration unless noted.
Recommended Supply
Use discount code PEPPAL at eligible peptide supplier checkouts.

Semaglutide Supply

SiPhox Health At-Home Blood Test
Injection Supplies
Disclosure: supply links may earn PDP a commission at no cost to you.
Peptide Vials (10 mg, 3.33 mg/mL mix)
A 10 mg vial reconstituted with 3.0 mL BAC water gives about 3.33 mg/mL. At a 2.4 mg weekly maintenance dose, one vial supports about 4 weeks. At 0.25 mg starter dose, one vial supports about 40 weeks. Plan on the maintenance dose, not the starter dose.
| Cycle length | Planning note |
|---|---|
8 weeks 1 vial | Covers full titration through 0.5 mg (Weeks 1-8 uses about 3 mg total; round up for losses). |
16 weeks 2 vials | Full titration to maintenance start. About 13.8 mg used through Week 16. |
24 weeks 4 vials | Adds 8 weeks at 2.4 mg maintenance (~33 mg total). Round up for priming losses. |
52 weeks 11 vials | Adds ~36 weeks at 2.4 mg (~100.2 mg). Round up. |
8 weeks
1 vial
Covers full titration through 0.5 mg (Weeks 1-8 uses about 3 mg total; round up for losses).
16 weeks
2 vials
Full titration to maintenance start. About 13.8 mg used through Week 16.
24 weeks
4 vials
Adds 8 weeks at 2.4 mg maintenance (~33 mg total). Round up for priming losses.
52 weeks
11 vials
Adds ~36 weeks at 2.4 mg (~100.2 mg). Round up.
Insulin Syringes (U-100)
Prefer 0.3 mL / 30-unit syringes during the 0.25-1.0 mg phase, and 1.0 mL / 100-unit syringes once doses cross 0.30 mL (30 units).
| Cycle length | Planning note |
|---|---|
8 weeks 8 syringes | 1 syringe per weekly injection. |
16 weeks 16 syringes | 1 per week. Buy a box of 100 to cover the full cycle. |
24 weeks 24 syringes | 1 per week. |
52 weeks 52 syringes | 1 per week. |
8 weeks
8 syringes
1 syringe per weekly injection.
16 weeks
16 syringes
1 per week. Buy a box of 100 to cover the full cycle.
24 weeks
24 syringes
1 per week.
52 weeks
52 syringes
1 per week.
Bacteriostatic Water
Use 3.0 mL per 10 mg vial for the standard 3.33 mg/mL mix.
| Cycle length | Planning note |
|---|---|
8-16 weeks 1 x 10 mL bottle | 8 weeks: 1 vial uses 3 mL.; 16 weeks: 2 vials use 6 mL. |
24 weeks 2 x 10 mL bottles | 4 vials use 12 mL. Two bottles give margin. |
52 weeks 4 x 10 mL bottles | 11 vials use 33 mL. Round up to 4 bottles for margin. |
8-16 weeks
1 x 10 mL bottle
8 weeks: 1 vial uses 3 mL.; 16 weeks: 2 vials use 6 mL.
24 weeks
2 x 10 mL bottles
4 vials use 12 mL. Two bottles give margin.
52 weeks
4 x 10 mL bottles
11 vials use 33 mL. Round up to 4 bottles for margin.
Round up for priming losses, dropped syringes, damaged swabs, and any titration delays. The math above assumes a 3.33 mg/mL mix on a 10 mg vial. Use the calculator for other vial sizes or concentrations.
Who Semaglutide Is For and Who Should Avoid It
Semaglutide is FDA-approved for several conditions, but it is not for everyone. Some people should avoid it entirely, and others need close medical oversight before starting.
Contraindications
- Personal or family history of medullary thyroid carcinoma (MTC). Semaglutide carries a boxed warning based on thyroid C-cell tumors in rodent studies.
- Multiple Endocrine Neoplasia syndrome type 2 (MEN 2).
- Known hypersensitivity to semaglutide or any ingredient in the formulation.
Use with caution
- Pregnancy and breastfeeding. Semaglutide is not approved for pregnancy. The label recommends stopping at least 2 months before a planned pregnancy because of the long half-life.
- History of pancreatitis. Pancreatitis has been reported in clinical trials. People with prior episodes need clinician oversight.
- Pre-existing diabetic retinopathy. In SUSTAIN-6, retinopathy complications were higher (3.0% vs 1.8% with placebo), likely tied to fast blood-sugar improvement in people who already had retinopathy.
- Gallbladder disease. Gallstones were more common with semaglutide (about 2.6% vs 1.2% with placebo) in the STEP trials, consistent with rapid weight loss in general.
- Severe gastrointestinal disease such as gastroparesis. Semaglutide already slows gastric emptying.
Not medical advice
Eligibility and safety decisions belong with a qualified clinician who knows your full medical history. This page is research reference only.
Semaglutide Side Effects & Safety
The most common side effects of semaglutide are stomach-related: nausea, diarrhea, vomiting, and constipation. They are usually worst during the first few months as the dose increases, and most people find they improve or go away with time. The safety profile below comes from tens of thousands of participants across the STEP, SUSTAIN, and SELECT trial programs.
Common stomach effects
In pooled STEP 1-3 data on the 2.4 mg dose, nausea occurred in 43.9% of participants (vs 16.1% on placebo), diarrhea in 29.7% (vs 15.9%), vomiting in 24.5% (vs 6.3%), and constipation in 24.2% (vs 11.1%). These events were most common during the first 16-20 weeks of escalation and largely passed with time. 99.5% of GI events were non-serious and 98.1% were mild-to-moderate.
Dose-dependent pattern
Lower diabetes doses (0.5-1.0 mg) produced stomach side effects in 32.7-36.4% of participants vs 15.3% on placebo. That is meaningfully lower than the 2.4 mg obesity dose. Higher doses give bigger effects but also more side effects.
Heart rate
Resting heart rate rose by about 1-4 beats per minute in clinical trials. Despite this small increase, SELECT showed a 20% drop in major adverse cardiovascular events in people with existing heart disease and obesity. No rise in heart failure hospitalizations was observed.
Gallbladder and pancreatitis
Gallstones were more common with semaglutide (about 2.6% vs 1.2% with placebo) in STEP, in line with rapid weight loss across all weight-loss interventions. Pancreatitis was rare but reported. Stop and seek care for severe abdominal pain.
Diabetic retinopathy
In SUSTAIN-6 (T2D), retinopathy complications were higher with semaglutide (3.0%) than placebo (1.8%). The most likely cause is fast blood sugar improvement in people who already had retinopathy.
Thyroid boxed warning
Semaglutide carries a boxed warning based on dose-dependent thyroid C-cell tumors in rodent studies. Human relevance is unknown. Semaglutide is contraindicated in anyone with a personal or family history of medullary thyroid carcinoma (MTC) or MEN 2.
Discontinuation
In STEP, 6.8% of people on semaglutide 2.4 mg stopped treatment because of side effects, vs 3.2% on placebo. The most common reasons were nausea (1.8%), vomiting (1.2%), and diarrhea (0.7%).
For a wider safety comparison, see the PepPal Peptide Side Effects Guide.
Semaglutide Timeline & What to Monitor
Semaglutide takes time to build up in the body and time to show meaningful effects. The 7-day half-life means blood levels keep rising for about 4-5 weeks at each dose before they stop changing.
Reasonable timing checkpoints based on clinical trial data
Checkpoint
Weeks 1-4 (0.25 mg)
What clinical trials show
Tolerability phase. No meaningful weight or HbA1c change expected.
Checkpoint
Weeks 5-8 (0.5 mg)
What clinical trials show
Early appetite changes possible. Most stomach side effects show up here.
Checkpoint
Weeks 8-12
What clinical trials show
Appetite suppression often clearly noticeable at 1.0 mg in diabetes research.
Checkpoint
Weeks 16-20
What clinical trials show
Stomach side effects usually plateau. Weight loss curve starts to accelerate.
Checkpoint
Week 28
What clinical trials show
STEP 1 interim data: significant weight loss vs placebo.
Checkpoint
Week 68
What clinical trials show
STEP 1 endpoint: 14.9% mean weight loss at 2.4 mg vs 2.4% placebo.
| Checkpoint | What clinical trials show |
|---|---|
| Weeks 1-4 (0.25 mg) | Tolerability phase. No meaningful weight or HbA1c change expected. |
| Weeks 5-8 (0.5 mg) | Early appetite changes possible. Most stomach side effects show up here. |
| Weeks 8-12 | Appetite suppression often clearly noticeable at 1.0 mg in diabetes research. |
| Weeks 16-20 | Stomach side effects usually plateau. Weight loss curve starts to accelerate. |
| Week 28 | STEP 1 interim data: significant weight loss vs placebo. |
| Week 68 | STEP 1 endpoint: 14.9% mean weight loss at 2.4 mg vs 2.4% placebo. |
These are clinical trial markers, not personal targets.
Markers studied in trials
- Body weight - weekly or monthly trend, not daily.
- HbA1c - 3-month average blood sugar. Used for type 2 diabetes research.
- Fasting glucose - short-term blood sugar marker.
- Blood pressure - SELECT showed small decreases.
- Resting heart rate - small increase (1-4 bpm) is normal.
- Lipid panel - mild improvements in LDL and triglycerides reported in trials.
- Kidney function (eGFR, UACR) - relevant if research touches CKD outcomes (FLOW).
- Liver enzymes - relevant if research touches MASH (ESSENCE).
Lab work belongs with a clinician
Ordering, interpreting, and acting on lab work is a clinical task. This list shows what trials track. It is not a personal monitoring plan.
Semaglutide Clinical Evidence Context
Semaglutide has been studied in more clinical trials than any other GLP-1 medication - over 45,000 participants across multiple programs. Each trial name (STEP, SELECT, FLOW, ESSENCE) is its own program. STEP tested weight loss. SUSTAIN and PIONEER tested diabetes control. SELECT tested heart protection. FLOW tested kidney protection. ESSENCE tested fatty liver disease (MASH). SOUL tested cardiovascular outcomes for oral semaglutide.
Major published semaglutide trials
Trial
STEP 1 (Wilding et al., NEJM 2021)
Design
Phase 3 / 68 weeks
Population
1,961 adults with obesity, no diabetes
Key result
-14.9% body weight (semaglutide 2.4 mg) vs -2.4% placebo. 86.4% reached 5%+ loss.
Trial
STEP 2 (Davies et al., Lancet 2021)
Design
Phase 3 / 68 weeks
Population
1,210 adults with T2D and overweight or obesity
Key result
-9.6% body weight (2.4 mg) vs -3.4% placebo.
Trial
STEP 3 (Wadden et al., JAMA 2021)
Design
Phase 3 / 68 weeks
Population
611 adults with obesity + intensive behavior therapy
Key result
-16.0% body weight (2.4 mg) vs -5.7% placebo.
Trial
STEP 5 (Garvey et al., Nature Med 2022)
Design
Phase 3 / 104 weeks
Population
304 adults with obesity, no diabetes
Key result
-15.2% body weight sustained at 2 years.
Trial
SELECT (Lincoff et al., NEJM 2023)
Design
Phase 3 / 39.8 mo mean follow-up
Population
17,604 adults with CVD + overweight or obesity, no diabetes
Key result
20% reduction in major heart events (HR 0.80; 95% CI 0.72-0.90; P<0.001). -9.4% body weight.
Trial
SUSTAIN-6 (Marso et al., NEJM 2016)
Design
Phase 3 / 104 weeks
Population
3,297 adults with T2D at high CV risk
Key result
26% MACE reduction (HR 0.74; 95% CI 0.58-0.95). First CV signal for semaglutide.
Trial
FLOW (Perkovic et al., NEJM 2024)
Design
Phase 3 / event-driven
Population
3,533 adults with T2D and CKD
Key result
24% reduction in kidney disease progression and CV death (HR 0.76; 95% CI 0.66-0.88).
Trial
ESSENCE (Sanyal et al., NEJM 2025)
Design
Phase 3 / 72 weeks (Part 1)
Population
Adults with MASH, F2-F3 fibrosis
Key result
33% achieved both steatohepatitis resolution and fibrosis improvement vs 16% placebo.
Trial
SOUL (McGuire et al., NEJM 2025)
Design
Phase 3 / event-driven
Population
Adults with T2D + ASCVD or CKD
Key result
Oral semaglutide significantly reduced MACE vs placebo in high-CV-risk T2D.
Trial
STEP TEENS (Weghuber et al., NEJM 2022)
Design
Phase 3 / 68 weeks
Population
201 adolescents (12-17) with obesity
Key result
-16.1% BMI reduction vs +0.6% placebo.
Trial
OASIS 4 (oral Wegovy)
Design
Phase 3 / 64 weeks
Population
Adults with obesity, no diabetes
Key result
-16.6% body weight at 25 mg daily. Basis for Dec 2025 oral Wegovy approval.
| Trial | Design | Population | Key result |
|---|---|---|---|
| STEP 1 (Wilding et al., NEJM 2021) | Phase 3 / 68 weeks | 1,961 adults with obesity, no diabetes | -14.9% body weight (semaglutide 2.4 mg) vs -2.4% placebo. 86.4% reached 5%+ loss. |
| STEP 2 (Davies et al., Lancet 2021) | Phase 3 / 68 weeks | 1,210 adults with T2D and overweight or obesity | -9.6% body weight (2.4 mg) vs -3.4% placebo. |
| STEP 3 (Wadden et al., JAMA 2021) | Phase 3 / 68 weeks | 611 adults with obesity + intensive behavior therapy | -16.0% body weight (2.4 mg) vs -5.7% placebo. |
| STEP 5 (Garvey et al., Nature Med 2022) | Phase 3 / 104 weeks | 304 adults with obesity, no diabetes | -15.2% body weight sustained at 2 years. |
| SELECT (Lincoff et al., NEJM 2023) | Phase 3 / 39.8 mo mean follow-up | 17,604 adults with CVD + overweight or obesity, no diabetes | 20% reduction in major heart events (HR 0.80; 95% CI 0.72-0.90; P<0.001). -9.4% body weight. |
| SUSTAIN-6 (Marso et al., NEJM 2016) | Phase 3 / 104 weeks | 3,297 adults with T2D at high CV risk | 26% MACE reduction (HR 0.74; 95% CI 0.58-0.95). First CV signal for semaglutide. |
| FLOW (Perkovic et al., NEJM 2024) | Phase 3 / event-driven | 3,533 adults with T2D and CKD | 24% reduction in kidney disease progression and CV death (HR 0.76; 95% CI 0.66-0.88). |
| ESSENCE (Sanyal et al., NEJM 2025) | Phase 3 / 72 weeks (Part 1) | Adults with MASH, F2-F3 fibrosis | 33% achieved both steatohepatitis resolution and fibrosis improvement vs 16% placebo. |
| SOUL (McGuire et al., NEJM 2025) | Phase 3 / event-driven | Adults with T2D + ASCVD or CKD | Oral semaglutide significantly reduced MACE vs placebo in high-CV-risk T2D. |
| STEP TEENS (Weghuber et al., NEJM 2022) | Phase 3 / 68 weeks | 201 adolescents (12-17) with obesity | -16.1% BMI reduction vs +0.6% placebo. |
| OASIS 4 (oral Wegovy) | Phase 3 / 64 weeks | Adults with obesity, no diabetes | -16.6% body weight at 25 mg daily. Basis for Dec 2025 oral Wegovy approval. |
ESSENCE Part 2 (240 weeks, liver outcomes) is ongoing, with results expected in 2029.
Semaglutide has the largest clinical evidence base of any GLP-1 receptor agonist. Its FDA label has grown from a single T2D indication in 2017 to six approved indications by early 2026: type 2 diabetes, chronic weight management (injectable and oral), cardiovascular risk reduction, chronic kidney disease protection, and MASH.
Semaglutide Storage & Handling
Storage by format
Format
Lyophilized (powder, sealed)
Storage
-4F (-20C) freezer
Shelf life
Up to 12+ months
Format
Lyophilized (powder, sealed)
Storage
35.6-46.4F (2-8C) refrigerator
Shelf life
Several months
Format
Lyophilized (powder, sealed)
Storage
Room temperature (shipping)
Shelf life
Stable for several weeks
Format
Reconstituted (solution)
Storage
35.6-46.4F (2-8C) refrigerator
Shelf life
Use within 28 days
Format
Reconstituted (solution)
Storage
-4F (-20C) freezer
Shelf life
Not recommended - do not freeze
| Format | Storage | Shelf life |
|---|---|---|
| Lyophilized (powder, sealed) | -4F (-20C) freezer | Up to 12+ months |
| Lyophilized (powder, sealed) | 35.6-46.4F (2-8C) refrigerator | Several months |
| Lyophilized (powder, sealed) | Room temperature (shipping) | Stable for several weeks |
| Reconstituted (solution) | 35.6-46.4F (2-8C) refrigerator | Use within 28 days |
| Reconstituted (solution) | -4F (-20C) freezer | Not recommended - do not freeze |
Brand pens (Ozempic, Wegovy) follow their own label storage rules.
- Protect reconstituted solutions from light.
- Do not freeze reconstituted semaglutide. Freezing can damage peptide integrity.
- Use bacteriostatic water (0.9% benzyl alcohol) for multi-dose vials. Sterile water is single-use only and should be discarded within 24 hours.
- Brand-name pens stay refrigerated before first use. After first use, pens may be stored at room temperature (up to 86F/30C) or refrigerated for up to 56 days per labeling.
Semaglutide Protocol Mistakes & Troubleshooting
Wrong BAC water volume
Adding the wrong amount of water changes the concentration and the syringe-unit math. For a 5 mg vial, 2.0 mL water = 2.5 mg/mL and 10 units = 0.25 mg. Adding 1.0 mL by mistake doubles the concentration to 5 mg/mL, so 10 units would then equal 0.5 mg - twice the intended starter dose. Recheck the math before drawing.
Cloudy or particulate vial
Reconstituted semaglutide should be clear and colorless with no visible particles. Cloudiness, color, or floating material can mean contamination or degradation. Discard the vial. Common causes: shaking during reconstitution, freeze-damage, or storage beyond 28 days after mixing.
Missed dose
If more than 2 days remain before the next scheduled weekly dose, the missed dose can be taken as soon as possible. If fewer than 2 days remain, skip it and resume the regular weekly schedule. Do not double up.
Bad nausea during escalation
Stomach side effects are most common during the 16-week escalation. FDA labeling allows staying at the current dose for an extra 4 weeks before stepping up. If the 2.4 mg maintenance dose is not tolerated, dropping back to 1.7 mg for 4 weeks before retrying is allowed.
Mixing up oral and injectable
Oral semaglutide doses (3-25 mg daily) are much larger than injectable doses (0.25-2.4 mg weekly) because absorption from the gut is very low (about 1%). The two routes are not interchangeable, and the milligram numbers do not translate directly.
Storage mistakes
Freezing reconstituted semaglutide can damage the peptide. So can letting reconstituted vials sit at room temperature for long stretches. Keep mixed vials at 2-8C and use within 28 days.
Semaglutide Regulatory Status
Semaglutide has one of the broadest FDA approval histories of any modern metabolic drug. As of May 2026, the approved indications are below.
FDA-approved semaglutide brands and indications
Brand
Ozempic (injectable)
Approval date
December 2017
Indication
Type 2 diabetes glycemic control
Brand
Ozempic (injectable)
Approval date
January 2020
Indication
Cardiovascular risk reduction in T2D
Brand
Rybelsus (oral tablet)
Approval date
September 2019
Indication
Type 2 diabetes (first oral GLP-1)
Brand
Wegovy (injectable)
Approval date
June 2021
Indication
Chronic weight management (BMI ≥30 or ≥27 with comorbidity)
Brand
Wegovy (injectable)
Approval date
March 2024
Indication
Cardiovascular risk reduction in adults with obesity and CVD
Brand
Wegovy (injectable)
Approval date
August 2025
Indication
Non-cirrhotic MASH with F2-F3 fibrosis (ESSENCE)
Brand
Wegovy oral 25 mg pill
Approval date
December 2025
Indication
Chronic weight management (OASIS 4 basis)
Brand
Ozempic (injectable)
Approval date
February 2026
Indication
CKD progression in T2D (FLOW basis)
| Brand | Approval date | Indication |
|---|---|---|
| Ozempic (injectable) | December 2017 | Type 2 diabetes glycemic control |
| Ozempic (injectable) | January 2020 | Cardiovascular risk reduction in T2D |
| Rybelsus (oral tablet) | September 2019 | Type 2 diabetes (first oral GLP-1) |
| Wegovy (injectable) | June 2021 | Chronic weight management (BMI ≥30 or ≥27 with comorbidity) |
| Wegovy (injectable) | March 2024 | Cardiovascular risk reduction in adults with obesity and CVD |
| Wegovy (injectable) | August 2025 | Non-cirrhotic MASH with F2-F3 fibrosis (ESSENCE) |
| Wegovy oral 25 mg pill | December 2025 | Chronic weight management (OASIS 4 basis) |
| Ozempic (injectable) | February 2026 | CKD progression in T2D (FLOW basis) |
Research-grade lyophilized semaglutide from supplier sources is not an FDA-approved drug product and is for research use only.
Generics
As of May 2026, no generic semaglutide is FDA-approved in the United States. Novo Nordisk holds the active patents on semaglutide. The FDA-recognized shortage of semaglutide ended in 2025, which limited the legal scope of 503A/503B compounded semaglutide. Some countries have different patent expirations; for example, generic semaglutide is expected to enter the Canadian market earlier than the United States. Verify the current status with FDA orange-book and country-specific regulators.
Semaglutide Cost & Access Context
Semaglutide cost varies a lot depending on the format and the source. Brand pens, compounded pharmacy semaglutide, and research-grade vials sit at very different price points.
Common semaglutide cost brackets (US, May 2026)
Source
Brand Wegovy / Ozempic
Format
Pre-filled pen
Typical cost band
$1,000-$1,400+ per month list price
Notes
Insurance coverage varies. Manufacturer savings cards exist for eligible patients.
Source
Brand Rybelsus
Format
Oral tablet
Typical cost band
Similar to brand pens per month
Notes
Daily dosing. Different mg numbers than injectable.
Source
Compounded pharmacy
Format
Vial or pen
Typical cost band
Highly variable
Notes
Legal scope narrowed after the FDA shortage ended in 2025. Verify the pharmacy is 503A or 503B compliant.
Source
Research-grade lyophilized vial
Format
5 mg or 10 mg vial
Typical cost band
Lower per-mg cost
Notes
Research use only. Not an FDA-approved drug product. Quality depends on the supplier's COA process.
| Source | Format | Typical cost band | Notes |
|---|---|---|---|
| Brand Wegovy / Ozempic | Pre-filled pen | $1,000-$1,400+ per month list price | Insurance coverage varies. Manufacturer savings cards exist for eligible patients. |
| Brand Rybelsus | Oral tablet | Similar to brand pens per month | Daily dosing. Different mg numbers than injectable. |
| Compounded pharmacy | Vial or pen | Highly variable | Legal scope narrowed after the FDA shortage ended in 2025. Verify the pharmacy is 503A or 503B compliant. |
| Research-grade lyophilized vial | 5 mg or 10 mg vial | Lower per-mg cost | Research use only. Not an FDA-approved drug product. Quality depends on the supplier's COA process. |
Prices change. Verify with current supplier and pharmacy quotes before planning.
For a wider supplier comparison and discount-code context, see the PepPal supplier rankings. For per-mg cost math, the PepPal calculator converts vial size and water volume into exact draws.
Semaglutide vs Tirzepatide vs Retatrutide vs Liraglutide
Semaglutide, tirzepatide, and retatrutide are the three most-discussed GLP-1-based compounds for weight management. They activate overlapping but different receptors, which leads to different efficacy levels, side effect profiles, and availability. Liraglutide is the older once-daily GLP-1 that semaglutide replaced for most uses.
Side-by-side comparison
Factor
Receptor targets
Semaglutide
GLP-1 only
Tirzepatide
GLP-1 + GIP
Retatrutide
GLP-1 + GIP + glucagon
Liraglutide
GLP-1 only
Factor
Half-life
Semaglutide
~7 days
Tirzepatide
~5 days
Retatrutide
~6 days
Liraglutide
~13 hours
Factor
Dosing frequency
Semaglutide
Once weekly
Tirzepatide
Once weekly
Retatrutide
Once weekly
Liraglutide
Once daily
Factor
Max studied dose
Semaglutide
2.4 mg/week (SubQ); 25 mg/day oral
Tirzepatide
15 mg/week
Retatrutide
12 mg/week
Liraglutide
3.0 mg/day
Factor
Peak trial weight loss
Semaglutide
-14.9% at 68 wks (STEP 1)
Tirzepatide
-22.5% at 72 wks (SURMOUNT-1)
Retatrutide
-28.7% at 68 wks (TRIUMPH-4)
Liraglutide
-8% at 56 wks (SCALE)
Factor
FDA status (May 2026)
Semaglutide
Approved (T2D, obesity, CVD, MASH, CKD, oral)
Tirzepatide
Approved (T2D, obesity)
Retatrutide
Investigational - Phase 3
Liraglutide
Approved (Saxenda obesity, Victoza T2D)
Factor
CV outcomes data
Semaglutide
Yes - SELECT (20% MACE drop)
Tirzepatide
SURPASS-CVOT ongoing
Retatrutide
TRIUMPH-3 ongoing
Liraglutide
Yes - LEADER (13% MACE drop)
| Factor | Semaglutide | Tirzepatide | Retatrutide | Liraglutide |
|---|---|---|---|---|
| Receptor targets | GLP-1 only | GLP-1 + GIP | GLP-1 + GIP + glucagon | GLP-1 only |
| Half-life | ~7 days | ~5 days | ~6 days | ~13 hours |
| Dosing frequency | Once weekly | Once weekly | Once weekly | Once daily |
| Max studied dose | 2.4 mg/week (SubQ); 25 mg/day oral | 15 mg/week | 12 mg/week | 3.0 mg/day |
| Peak trial weight loss | -14.9% at 68 wks (STEP 1) | -22.5% at 72 wks (SURMOUNT-1) | -28.7% at 68 wks (TRIUMPH-4) | -8% at 56 wks (SCALE) |
| FDA status (May 2026) | Approved (T2D, obesity, CVD, MASH, CKD, oral) | Approved (T2D, obesity) | Investigational - Phase 3 | Approved (Saxenda obesity, Victoza T2D) |
| CV outcomes data | Yes - SELECT (20% MACE drop) | SURPASS-CVOT ongoing | TRIUMPH-3 ongoing | Yes - LEADER (13% MACE drop) |
These compounds are not interchangeable. Dose ranges, reconstitution math, and titration schedules all differ.
What sets each apart
- Semaglutide has the deepest evidence base across diabetes, obesity, heart, kidney, MASH, and oral. No other GLP-1 has six approved indications.
- Tirzepatide produced greater weight loss head-to-head: -20.2% vs -13.7% for semaglutide 2.4 mg in SURMOUNT-5.
- Retatrutide has the highest observed weight loss to date but is still investigational. Glucagon agonism adds energy expenditure and liver-fat reduction signals.
- Liraglutide is older and less effective for weight loss but has long real-world history and may still fit specific clinical scenarios.
For non-GLP-1 metabolic alternatives, see the tesofensine protocol (triple monoamine reuptake inhibitor) or the MOTS-c protocol (mitochondrial peptide).
Semaglutide Blood Tests & Monitoring
Semaglutide is an approved GLP-1 receptor agonist used in diabetes and weight-management contexts. Monitoring focuses on glucose control, kidney context, lipids, GI symptoms, gallbladder/pancreas symptoms, and thyroid-history cautions.
Blood test markers to discuss with a clinician
Marker
A1c
Why it matters
Shows longer-term glucose control and helps track metabolic response over time.
Timing
Baseline
Marker
Fasting glucose
Why it matters
Gives a current glucose snapshot, especially when appetite and diabetes medication needs change.
Timing
Follow-up
Marker
Comprehensive metabolic panel (CMP)
Why it matters
Reviews kidney function, liver enzymes, electrolytes, and glucose, which matter with GI symptoms or dehydration.
Timing
Baseline
Marker
Lipid panel
Why it matters
Tracks cardiometabolic changes during weight-loss or diabetes-management phases.
Timing
Follow-up
Marker
Blood pressure and resting heart rate
Why it matters
Adds cardiovascular context during weight loss, medication changes, and hydration shifts.
Timing
Optional
| Marker | Why it matters | Timing |
|---|---|---|
| A1c | Shows longer-term glucose control and helps track metabolic response over time. | Baseline |
| Fasting glucose | Gives a current glucose snapshot, especially when appetite and diabetes medication needs change. | Follow-up |
| Comprehensive metabolic panel (CMP) | Reviews kidney function, liver enzymes, electrolytes, and glucose, which matter with GI symptoms or dehydration. | Baseline |
| Lipid panel | Tracks cardiometabolic changes during weight-loss or diabetes-management phases. | Follow-up |
| Blood pressure and resting heart rate | Adds cardiovascular context during weight loss, medication changes, and hydration shifts. | Optional |
Monitoring guidance is label-informed for semaglutide and supported by GLP-1 clinical monitoring considerations.
At-home blood test option
Easy at home option to monitor core metrics during research cycles.

Partner link: PDP may earn a commission at no cost to you.
Simple timing framework
Baseline
Discuss baseline labs before starting or changing dose, especially with diabetes, kidney disease, pancreatitis history, gallbladder history, thyroid cancer history, or major calorie restriction.
Follow-up
Repeat glucose and CMP-related markers after 4-12 weeks or sooner if vomiting, dehydration, or glucose symptoms occur.
Longer term
For ongoing use, review metabolic and kidney trends every 3-6 months with a clinician.
How to interpret the labs
- Diabetes medications may need clinician review if glucose drops with reduced intake.
- Severe abdominal pain, gallbladder symptoms, and persistent vomiting require symptom-based review.
- Personal or family history of medullary thyroid carcinoma or MEN2 is an important label-based caution.
Do not wait for routine labs
Severe abdominal pain, persistent vomiting, signs of dehydration, fainting, allergic symptoms, or severe low-glucose symptoms need medical review. A neck mass, trouble swallowing, or persistent hoarseness should be discussed with a clinician.
FAQ
Q1: What is the starting dose of semaglutide?
The FDA-approved starting dose for weight management (Wegovy) is 0.25 mg once weekly by subcutaneous injection. This dose is for tolerability only and is not considered therapeutic. After 4 weeks at 0.25 mg, the dose increases to 0.5 mg weekly, then steps up every 4 weeks through 1.0 mg and 1.7 mg until reaching the 2.4 mg maintenance dose by about week 17. For type 2 diabetes (Ozempic), the same 0.25 mg starting dose applies, but the maximum dose is 2.0 mg weekly.
Q2: What is semaglutide's half-life?
Semaglutide has a half-life of about 7 days (165-184 hours), among the longest for current GLP-1 receptor agonists. A fatty acid chain attached to the peptide binds to albumin in the blood, which slows clearance. The 7-day half-life supports once-weekly dosing and means it takes about 4-5 weeks at each dose level to reach steady state. After stopping, semaglutide remains in the body for roughly 5-7 weeks.
Q3: How much weight can you lose on semaglutide?
In the STEP 1 clinical trial (NEJM, 2021), participants on semaglutide 2.4 mg lost an average of 14.9% of body weight at 68 weeks (about 33.6 lbs from a 232 lb average baseline) vs 2.4% with placebo. 86.4% achieved at least 5% loss, and about one-third reached 20% or more. STEP 3 with intensive behavior therapy showed 16.0% loss. STEP 5 showed sustained 15.2% loss at 2 years. STEP 2 in type 2 diabetes showed 9.6% loss.
Q4: How do you reconstitute semaglutide?
Reconstitution applies to lyophilized (freeze-dried) research-grade semaglutide. Brand Ozempic and Wegovy pens come pre-filled and skip this step. A common setup is a 5 mg vial with 2.0 mL bacteriostatic water, which yields a 2.5 mg/mL concentration where 0.25 mg equals 10 units on a U-100 syringe. Inject water slowly down the vial wall, swirl gently (do not shake), and refrigerate at 2-8C. Use within 28 days. For custom math, use the Pep Pal calculator.
Q5: Is semaglutide FDA-approved?
Yes. As of May 2026, semaglutide has broad FDA approval across multiple indications and brand names. Ozempic is approved for type 2 diabetes glycemic control (2017), cardiovascular risk reduction in T2D (2020), and CKD progression in T2D (February 2026). Wegovy is approved for chronic weight management (2021), cardiovascular risk reduction in adults with obesity and CVD (2024), and non-cirrhotic MASH with F2-F3 fibrosis (August 2025). The oral Wegovy 25 mg pill was approved in December 2025. Rybelsus is the older oral T2D form.
Q6: What are the most common side effects?
Stomach side effects are most common. In pooled STEP 1-3 data on the 2.4 mg dose, nausea occurred in 43.9% (vs 16.1% placebo), diarrhea in 29.7% (vs 15.9%), vomiting in 24.5% (vs 6.3%), and constipation in 24.2% (vs 11.1%). These were most frequent during dose escalation, peaked around weeks 8-20, and were usually mild-to-moderate and transient. Permanent discontinuation due to side effects was 6.8%. Semaglutide carries a boxed warning for thyroid C-cell tumors in rodent studies and is contraindicated in people with MTC or MEN 2.
Q7: How does semaglutide compare to tirzepatide and retatrutide?
Semaglutide is a single GLP-1 receptor agonist. Tirzepatide is a dual GLP-1 + GIP agonist. Retatrutide is a triple GLP-1 + GIP + glucagon agonist. In SURMOUNT-5 head-to-head, tirzepatide 15 mg produced -20.2% weight loss vs -13.7% for semaglutide 2.4 mg at 72 weeks. Retatrutide Phase 3 reported up to -28.7% at 68 weeks. Semaglutide's strength is depth of outcomes evidence: SELECT cardiovascular data, FLOW kidney protection, MASH approval (ESSENCE), and oral formulations.
Q8: What vial sizes are available for research semaglutide?
Lyophilized semaglutide is commonly offered by research suppliers in 2 mg, 3 mg, 5 mg, and 10 mg vials. The 5 mg vial is the most common pick because it supports multiple weeks at typical doses and pairs cleanly with 2.0 mL bacteriostatic water for a 2.5 mg/mL mix. The 10 mg vial can be more cost-effective for higher-dose or longer protocols. Vial size and water volume should be chosen together, because they drive the syringe-unit math.
Q9: How much bacteriostatic water should be added to semaglutide?
BAC water volume depends on vial size and desired concentration. For a 5 mg vial, 2.0 mL gives 2.5 mg/mL, where 0.25 mg = 10 units and 2.4 mg = 96 units on a U-100 syringe. For a 10 mg vial, 3.0 mL gives about 3.33 mg/mL, where 0.25 mg = 7.5 units and 2.4 mg = 72 units. A more concentrated 5 mg/mL option (5 mg in 1.0 mL) gives smaller draws but needs more precise syringe handling. Use the Pep Pal calculator for any custom setup.
Q10: What is the maximum dose of semaglutide studied?
The maximum FDA-approved subcutaneous dose is 2.4 mg weekly (Wegovy) for weight management. For type 2 diabetes (Ozempic), the maximum approved dose is 2.0 mg weekly. The oral maximum is 25 mg daily (Wegovy 25 mg pill, approved December 2025) or 14 mg daily (Rybelsus). In SELECT, the 2.4 mg dose produced a 20% reduction in major cardiovascular events. Published Phase 3 programs have not established higher subcutaneous maintenance doses than 2.4 mg weekly.
Q11: How should reconstituted semaglutide be stored?
Reconstituted semaglutide should be refrigerated at 2-8C (35.6-46.4F) and used within 28 days. Do not freeze reconstituted semaglutide - freezing can degrade the peptide. Protect from light and label vials with the reconstitution date and concentration. Brand-name pens follow their own label rules: unused pens stay refrigerated, and after first use, pens can be stored at room temperature (up to 86F/30C) or refrigerated for up to 56 days.
Q12: Is Wegovy 25 mg the same as Rybelsus?
No. Both are oral semaglutide, but they are different products with different maximum doses and approved indications. Rybelsus tops out at 14 mg daily and is approved for type 2 diabetes. The Wegovy 25 mg pill was approved in December 2025 for chronic weight management and tops out at 25 mg daily. Both must be taken on an empty stomach with a small sip of water, with no food, drink, or other medication for 30 minutes after.
Q13: Is semaglutide insulin?
No. Semaglutide is not insulin. It is a GLP-1 receptor agonist - a drug that copies the natural gut hormone GLP-1. It does cause the pancreas to release insulin, but only when blood sugar is already high (glucose-dependent insulin secretion). That is why semaglutide is much less likely to cause dangerous low blood sugar than direct insulin. Insulin works by directly adding the hormone insulin to the body. Semaglutide works by signaling the body to release its own insulin when it is needed.
Q14: Is there a generic semaglutide?
As of May 2026, no generic semaglutide is FDA-approved in the United States. Novo Nordisk still holds active patents. The FDA-recognized shortage of semaglutide ended in 2025, which narrowed the legal scope of 503A/503B compounded semaglutide. Some countries have different patent timelines, with generic semaglutide expected in Canada earlier than the US. Verify current status with FDA orange-book listings and country-specific regulators before relying on this answer.
Q15: Where can I calculate reconstitution and syringe units?
Use the Pep Pal calculator for exact dose-to-unit conversions. Enter vial size, BAC water volume, and target dose to get the matching syringe draw.
Q16: Where can I compare peptide suppliers?
Browse the PepPal supplier directory for current supplier listings, or read the PepPal supplier rankings for ranked comparisons with Finnrick testing context.
Sources & Research
- 1. Wilding JPH, Batterham RL, Calanna S, et al. Once-Weekly Semaglutide in Adults with Overweight or Obesity (STEP 1). New England Journal of Medicine (2021)
- 2. Lincoff AM, Brown-Frandsen K, Colhoun HM, et al. Semaglutide and Cardiovascular Outcomes in Obesity without Diabetes (SELECT). New England Journal of Medicine (2023)
- 3. Marso SP, Bain SC, Consoli A, et al. Semaglutide and Cardiovascular Outcomes in Patients with Type 2 Diabetes (SUSTAIN-6). New England Journal of Medicine (2016)
- 4. Perkovic V, Tuttle KR, Rossing P, et al. Effects of Semaglutide on Chronic Kidney Disease in Patients with Type 2 Diabetes (FLOW). New England Journal of Medicine (2024)
- 5. Sanyal AJ, Newsome PN, Kliers I, et al. Phase 3 Trial of Semaglutide in Metabolic Dysfunction-Associated Steatohepatitis (ESSENCE). New England Journal of Medicine (2025)
- 6. Wharton S, Calanna S, Davies M, et al. Gastrointestinal Tolerability of Once-Weekly Semaglutide 2.4 mg in Adults with Overweight or Obesity. Diabetes, Obesity and Metabolism (2022)
- 7. Weghuber D, Barrett T, Barrientos-Perez M, et al. Once-Weekly Semaglutide in Adolescents with Obesity (STEP TEENS). New England Journal of Medicine (2022)
- 8. McGuire DK, et al. Oral Semaglutide and Cardiovascular Outcomes in High-Risk Type 2 Diabetes (SOUL). New England Journal of Medicine (2025)
- 9. Davies M, Faerch L, Jeppesen OK, et al. Semaglutide 2.4 mg Once a Week in Adults with Overweight or Obesity and Type 2 Diabetes (STEP 2). Lancet (2021)
- 10. U.S. Food and Drug Administration. Wegovy (semaglutide) injection - Prescribing Information. FDA (2025)
- 11. U.S. Food and Drug Administration. Ozempic (semaglutide) injection - Prescribing Information. FDA (2025)
- 12. ClinicalTrials.gov. STEP 1: Research Study Investigating How Well Semaglutide Works in People Suffering from Overweight or Obesity (NCT03548935). ClinicalTrials.gov (2018)
Related Dosing Protocols
Educational use only
This guide is an educational research reference, not medical advice or a treatment plan. Semaglutide is FDA-approved under Ozempic, Wegovy, and Rybelsus for specific indications. Research-grade lyophilized semaglutide discussed in reconstitution sections is for research use only. Consult a qualified clinician before considering any peptide protocol.
Calculate vial math
Use the calculator for custom vial size, BAC water volume, and syringe-unit math.
Open CalculatorWritten by Garret Grant
Founder & Lead Researcher · B.S. Civil Engineering, UCLA
Last updated: May 2026
Human-researched and AI-assisted with full editorial review. I verify sources, protocol interpretation, and final judgments personally. See methodology.
Share this page
