Cagrilintide Quick Start
Cagrilintide is a once-weekly research peptide developed by Novo Nordisk. It is a long-acting analogue of amylin, the hormone the pancreas releases with insulin to signal fullness after eating. In plain English, cagrilintide is studied because it is designed to make people feel full sooner, stay full longer, and slow how fast the stomach empties.
This page focuses on cagrilintide-only research planning. The common standalone ladder starts at 0.25 mg once weekly, escalates every 4 weeks, and reaches a 2.4 mg weekly research-target dose from week 17 onward. The cagrilintide + semaglutide combination is covered separately on the CagriSema stack page.
Route
Subcutaneous injection, once weekly, on the same day each week.
Half-life
About 159 to 195 hours (~7 to 8 days), which is why it is dosed weekly.
Vial sizes
Most research suppliers list 5 mg or 10 mg lyophilized vials.
Reconstitute
BAC water added per vial; concentration depends on volume chosen.
Research status
Phase 3 complete for monotherapy and CagriSema; standalone not FDA-approved.
Disclaimer
This page is an educational research reference and is not medical advice. Cagrilintide is investigational. No content here should be interpreted as a treatment recommendation or as a substitute for qualified clinical care.
This page helps with dose planning, vial math, timing, and storage. For the wider research view, see the Cagrilintide guide on how it works, study results, side effects, and legal status.
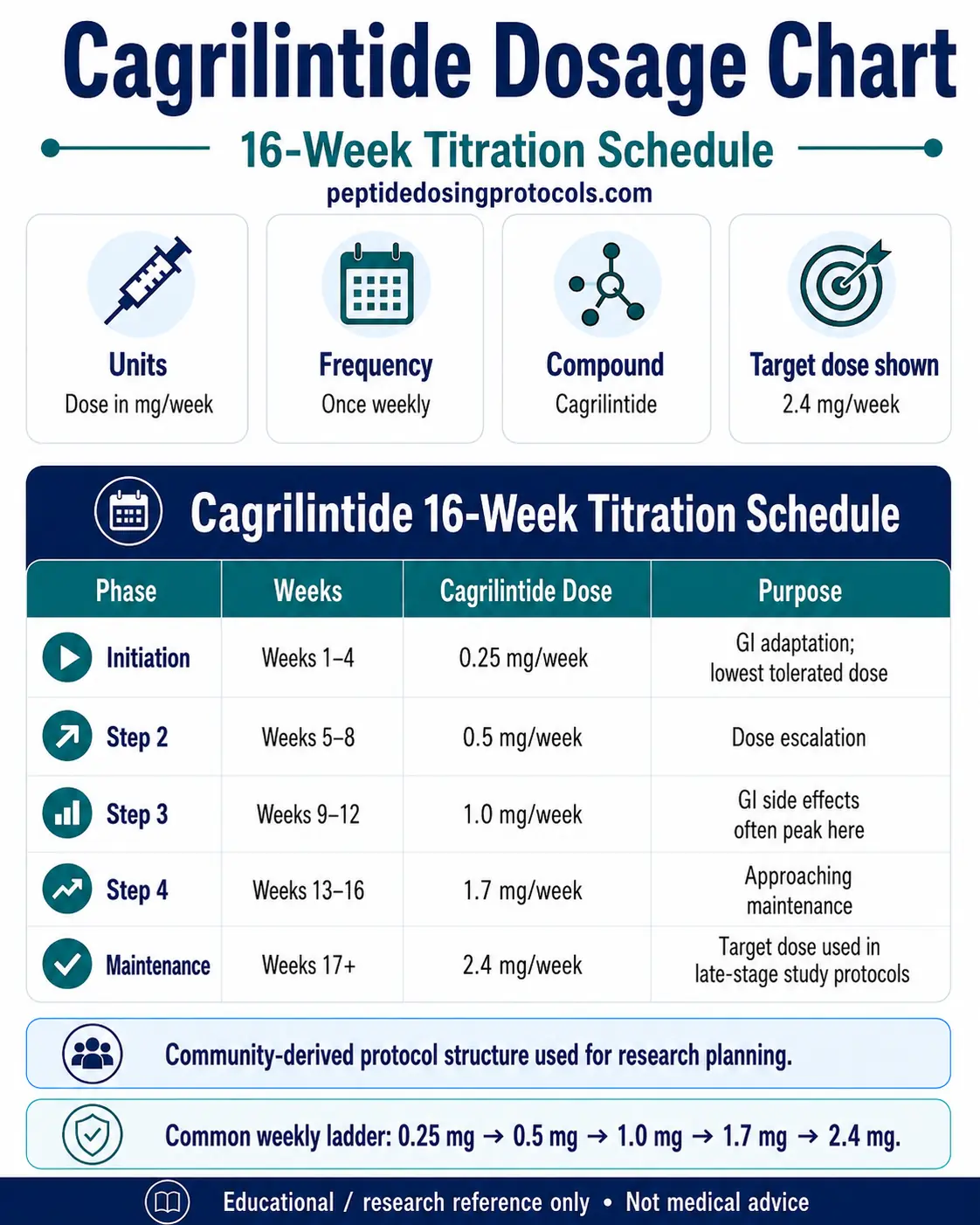
Cagrilintide Dosing Protocol & Schedule
The cagrilintide-only dosing schedule below keeps the amylin analogue separate from CagriSema combination dosing. It uses a 16-week escalation ladder so tolerability can be reviewed before the 2.4 mg weekly research-target dose.
Cagrilintide 16-Week Titration Schedule
Phase
Initiation
Weeks
Weeks 1-4
Cagrilintide Dose
0.25 mg/week
Purpose
GI adaptation; lowest tolerated dose
Phase
Step 2
Weeks
Weeks 5-8
Cagrilintide Dose
0.5 mg/week
Purpose
Dose escalation
Phase
Step 3
Weeks
Weeks 9-12
Cagrilintide Dose
1.0 mg/week
Purpose
GI side effects often peak here
Phase
Step 4
Weeks
Weeks 13-16
Cagrilintide Dose
1.7 mg/week
Purpose
Approaching maintenance
Phase
Maintenance
Weeks
Weeks 17+
Cagrilintide Dose
2.4 mg/week
Purpose
Target dose used in late-stage study protocols
| Phase | Weeks | Cagrilintide Dose | Purpose |
|---|---|---|---|
| Initiation | Weeks 1-4 | 0.25 mg/week | GI adaptation; lowest tolerated dose |
| Step 2 | Weeks 5-8 | 0.5 mg/week | Dose escalation |
| Step 3 | Weeks 9-12 | 1.0 mg/week | GI side effects often peak here |
| Step 4 | Weeks 13-16 | 1.7 mg/week | Approaching maintenance |
| Maintenance | Weeks 17+ | 2.4 mg/week | Target dose used in late-stage study protocols |
Each step lasts 4 weeks. Researchers commonly hold a step longer if GI side effects are not tolerated rather than skip ahead. This cagrilintide-only table does not include semaglutide.
Earlier Phase 2 monotherapy work also studied higher weekly doses up to 4.5 mg, but the cagrilintide-only schedule above matches the 2.4 mg weekly target shown throughout this page. For the cagrilintide + semaglutide combination, see the CagriSema stack.
Cycle Guidelines
Common Cagrilintide Research Cycle Lengths
Approach
Short cycle
Duration
16 weeks
Review Point
Week 16
Notes
Reaches maintenance just as titration completes
Approach
Standard cycle
Duration
26-32 weeks
Review Point
Week 26 / 32
Notes
Matches Phase 2 trial durations
Approach
Long cycle
Duration
68 weeks
Review Point
Week 68
Notes
Matches REDEFINE 1 and REDEFINE 2 endpoints
| Approach | Duration | Review Point | Notes |
|---|---|---|---|
| Short cycle | 16 weeks | Week 16 | Reaches maintenance just as titration completes |
| Standard cycle | 26-32 weeks | Week 26 / 32 | Matches Phase 2 trial durations |
| Long cycle | 68 weeks | Week 68 | Matches REDEFINE 1 and REDEFINE 2 endpoints |
Cycle length is a research planning choice, not a treatment recommendation. Longer cycles match the published trial endpoints used to estimate weight-loss outcomes.
Missed dose: Because cagrilintide has a 7- to 8-day half-life, the published label-style guidance is to take a missed dose as soon as possible if the next scheduled dose is more than 3 days away, or to skip the missed dose if the next dose is closer than 3 days. Do not double dose.
Need exact units for a custom vial size, BAC volume, or dose? Use the PepPal cagrilintide reconstitution calculator.
Cagrilintide Supplies Needed
Plan supplies based on cycle length and the dosing schedule above. The vial math below assumes a 5 mg vial reconstituted with 2.0 mL BAC water (2.5 mg/mL) for the 2.4 mg/week target. Adjust if using a 10 mg vial or different concentration.
Recommended Supply
PEPPAL applies to eligible Peptide Partners checkout links.

Cagrilintide (10mg vials X 5)

Cagrilintide (10mg vial)

SiPhox Health At-Home Blood Test
Injection Supplies
Disclosure: supply links may earn PDP a commission at no cost to you.
Peptide Vials (5 mg vial, 2.4 mg/week target)
Each 5 mg vial covers about 2 weekly doses at 2.4 mg/week. Round up to leave priming margin.
| Cycle length | Planning note |
|---|---|
16 weeks 9 vials | Titration uses less; maintenance phase is the heavy draw |
26 weeks 13 vials | Standard Phase 2 duration |
32 weeks 16 vials | Phase 2 T2D study duration |
68 weeks 30 vials | REDEFINE 1 / REDEFINE 2 endpoint duration |
16 weeks
9 vials
Titration uses less; maintenance phase is the heavy draw
26 weeks
13 vials
Standard Phase 2 duration
32 weeks
16 vials
Phase 2 T2D study duration
68 weeks
30 vials
REDEFINE 1 / REDEFINE 2 endpoint duration
Peptide Vials (10 mg vial, 2.4 mg/week target)
Each 10 mg vial covers about 4 weekly doses at 2.4 mg/week. Round up for priming margin.
| Cycle length | Planning note |
|---|---|
16 weeks 5 vials | Lower vial count but larger draw volumes per dose |
26 weeks 7 vials | Standard Phase 2 duration |
32 weeks 8 vials | Phase 2 T2D study duration |
68 weeks 17 vials | Long-cycle planning matches REDEFINE 1 and 2 |
16 weeks
5 vials
Lower vial count but larger draw volumes per dose
26 weeks
7 vials
Standard Phase 2 duration
32 weeks
8 vials
Phase 2 T2D study duration
68 weeks
17 vials
Long-cycle planning matches REDEFINE 1 and 2
Insulin Syringes (U-100, 1 mL)
One syringe per weekly injection. 1 mL syringe is preferred at 2.4 mg with 2.5 mg/mL since 0.5 mL syringes cap below the 0.96 mL draw.
| Cycle length | Planning note |
|---|---|
16 weeks 16 syringes | 1 syringe per weekly injection |
26 weeks 26 syringes | 1 syringe per weekly injection |
32 weeks 32 syringes | 1 syringe per weekly injection |
68 weeks 68 syringes | 1 box of 100 covers a long cycle |
16 weeks
16 syringes
1 syringe per weekly injection
26 weeks
26 syringes
1 syringe per weekly injection
32 weeks
32 syringes
1 syringe per weekly injection
68 weeks
68 syringes
1 box of 100 covers a long cycle
Bacteriostatic Water (10 mL bottles)
Each 5 mg vial uses 2.0 mL BAC water; each 10 mg vial typically uses 3.0 mL.
| Cycle length | Planning note |
|---|---|
16 weeks 1 x 10 mL bottle | Covers 4-5 vials per bottle |
26-32 weeks 2 x 10 mL bottles | Margin for priming losses |
68 weeks 4 x 10 mL bottles | Long-cycle margin |
16 weeks
1 x 10 mL bottle
Covers 4-5 vials per bottle
26-32 weeks
2 x 10 mL bottles
Margin for priming losses
68 weeks
4 x 10 mL bottles
Long-cycle margin
Round up for priming losses, dropped syringes, damaged swabs, and any protocol adjustments. If using monotherapy at 4.5 mg/week, vial counts roughly double from the 2.4 mg/week math above.
Cagrilintide Reconstitution Guide
Reconstitution is the process of mixing the lyophilized cagrilintide powder with bacteriostatic water (BAC water) so it becomes injectable. The volume of BAC water added decides the final concentration in mg/mL, which in turn decides the syringe draw volume for any target dose.
Common Cagrilintide Reconstitution Setups
Vial Size
5 mg
BAC Water
2.0 mL
Concentration
2.5 mg/mL
0.6 mg Draw
0.24 mL / 24 units
1.2 mg Draw
0.48 mL / 48 units
2.4 mg Draw
0.96 mL / 96 units
4.5 mg Draw
1.80 mL / 180 units*
Vial Size
5 mg
BAC Water
3.0 mL
Concentration
1.67 mg/mL
0.6 mg Draw
0.36 mL / 36 units
1.2 mg Draw
0.72 mL / 72 units
2.4 mg Draw
1.44 mL / 144 units*
4.5 mg Draw
Exceeds vial
Vial Size
10 mg
BAC Water
2.0 mL
Concentration
5.0 mg/mL
0.6 mg Draw
0.12 mL / 12 units
1.2 mg Draw
0.24 mL / 24 units
2.4 mg Draw
0.48 mL / 48 units
4.5 mg Draw
0.90 mL / 90 units
Vial Size
10 mg
BAC Water
3.0 mL
Concentration
3.33 mg/mL
0.6 mg Draw
0.18 mL / 18 units
1.2 mg Draw
0.36 mL / 36 units
2.4 mg Draw
0.72 mL / 72 units
4.5 mg Draw
1.35 mL / 135 units*
| Vial Size | BAC Water | Concentration | 0.6 mg Draw | 1.2 mg Draw | 2.4 mg Draw | 4.5 mg Draw |
|---|---|---|---|---|---|---|
| 5 mg | 2.0 mL | 2.5 mg/mL | 0.24 mL / 24 units | 0.48 mL / 48 units | 0.96 mL / 96 units | 1.80 mL / 180 units* |
| 5 mg | 3.0 mL | 1.67 mg/mL | 0.36 mL / 36 units | 0.72 mL / 72 units | 1.44 mL / 144 units* | Exceeds vial |
| 10 mg | 2.0 mL | 5.0 mg/mL | 0.12 mL / 12 units | 0.24 mL / 24 units | 0.48 mL / 48 units | 0.90 mL / 90 units |
| 10 mg | 3.0 mL | 3.33 mg/mL | 0.18 mL / 18 units | 0.36 mL / 36 units | 0.72 mL / 72 units | 1.35 mL / 135 units* |
*Draws above 1.0 mL exceed standard 1 mL U-100 syringe capacity and may require a larger syringe or split injections. Units shown for U-100 insulin syringes.
7-Step Reconstitution Process
- 01
Bring vial to room temperature
Remove the lyophilized cagrilintide vial from cold storage and let it sit for 10-15 minutes. Do not heat or microwave.
- 02
Clean the stopper
Wipe the rubber stopper with an alcohol swab and let it air dry completely before piercing.
- 03
Draw BAC water
Using a sterile syringe, draw the planned BAC water volume (commonly 2.0 mL or 3.0 mL).
- 04
Inject slowly down the wall
Insert the needle at a slight angle and let the BAC water trickle slowly down the inside wall of the vial. Do not spray it directly onto the powder.
- 05
Swirl gently
Roll or swirl the vial gently in your palms until the powder fully dissolves into a clear solution. Do not shake.
- 06
Inspect for clarity
Look for any cloudiness, color, or particles. Discard the vial if any are present.
- 07
Label and refrigerate
Write the reconstitution date and concentration on the vial. Store at 2-8 degrees Celsius (35.6-46.4 degrees Fahrenheit) and use within 28-30 days.
Need exact syringe units?
Use the PepPal cagrilintide reconstitution calculator for any vial size, BAC volume, or target dose. Doses over 1.0 mL exceed the standard 1 mL U-100 syringe.
Cagrilintide Dosage Chart
This cagrilintide dosage chart summarizes the standalone 16-week titration ladder from 0.25 mg weekly to the 2.4 mg weekly research-target dose.
How Cagrilintide Works
Cagrilintide mimics amylin, a hormone the pancreas releases with insulin to tell the brain that the body is full. The compound is engineered to last about 7-8 days in the body so it can be dosed once a week, instead of needing multiple injections like the natural hormone.
Pharmacologically, cagrilintide is a dual amylin and calcitonin receptor agonist (DACRA) — meaning it activates two related receptor systems at once.
Amylin Receptors (Brain): Satiety and Appetite
Amylin receptors are concentrated in brain regions that handle fullness signals, including the area postrema and hypothalamus. When cagrilintide activates them, appetite drops and the sensation of fullness lasts longer between meals. Animal evidence suggests this pathway also helps preserve lean mass during weight reduction, although the human data is still maturing.
Calcitonin Receptor (Gut): Slowed Gastric Emptying
The calcitonin receptor adds a second effect. It slows gastric emptying — meaning food leaves the stomach more slowly — which extends post-meal fullness. It is also linked to lower glucagon release after meals, which can improve glycemic stability.
Why Cagrilintide Pairs With GLP-1 Drugs
GLP-1 drugs (semaglutide, tirzepatide) act on a different receptor system that overlaps but is not identical to amylin signaling. Stacking the two creates additive appetite suppression rather than redundant signaling. This is the pharmacological basis for CagriSema: in REDEFINE 1, the combination produced 20.4% mean weight loss versus 11.8% for cagrilintide alone and 14.9% for semaglutide alone.
Who Cagrilintide Is For and Who Should Avoid It
Cagrilintide is investigational. It is studied in adults with overweight or obesity (BMI 27 or higher in REDEFINE 1) and in adults with type 2 diabetes (REDEFINE 2 and REIMAGINE 2). The published trials excluded several populations and conditions; that exclusion list is the most useful guide to who research planning should not include.
Excluded or Cautioned Populations in Phase 3 Trials
Phase 3 REDEFINE/REIMAGINE protocols typically excluded participants with: Type 1 diabetes; history of pancreatitis; severe gastroparesis; pregnancy or breastfeeding; recent major cardiovascular events; active malignancy; severe renal or hepatic impairment; personal or family history of medullary thyroid carcinoma or MEN2 (a class-level GLP-1 caution that may extend to combination products).
Medication Conflicts
Because cagrilintide slows gastric emptying, it can change how oral medications are absorbed. Drugs with narrow therapeutic windows (warfarin, levothyroxine, oral contraceptives, certain seizure medications) deserve clinician oversight. Insulin and other glucose-lowering drugs may need adjustment to reduce hypoglycemia risk.
Clinician oversight
Cagrilintide is not approved for individual self-administration. Anyone with metabolic, cardiac, GI, hepatic, or renal disease — or who is pregnant, breastfeeding, or planning pregnancy — should not consider it without licensed clinical care.
Cagrilintide Side Effects & Safety
Most side effects reported in cagrilintide trials are gastrointestinal and dose-dependent. They typically peak during dose escalation and ease as the body adapts to a stable dose.
Observed Side Effects (From Clinical Trials)
In the Lancet 2021 Phase 2 trial (n=706), at the highest 4.5 mg/week dose, 88% of participants reported at least one side effect and 63% reported a GI-specific event. Across all doses, GI events were reported in 41-63% of cagrilintide groups versus 32% with placebo.
Phase 2 Side-Effect Frequencies at 4.5 mg/week (Lancet 2021)
Side Effect
Nausea
Incidence at 4.5 mg/week
47%
Side Effect
Injection-site reactions
Incidence at 4.5 mg/week
43%
Side Effect
Constipation
Incidence at 4.5 mg/week
21%
Side Effect
Fatigue
Incidence at 4.5 mg/week
20%
Side Effect
Allergic reactions (any)
Incidence at 4.5 mg/week
10%
Side Effect
Vomiting
Incidence at 4.5 mg/week
8%
Side Effect
Loose stools
Incidence at 4.5 mg/week
7%
Side Effect
Headache
Incidence at 4.5 mg/week
7%
Side Effect
Indigestion
Incidence at 4.5 mg/week
4%
| Side Effect | Incidence at 4.5 mg/week |
|---|---|
| Nausea | 47% |
| Injection-site reactions | 43% |
| Constipation | 21% |
| Fatigue | 20% |
| Allergic reactions (any) | 10% |
| Vomiting | 8% |
| Loose stools | 7% |
| Headache | 7% |
| Indigestion | 4% |
Incidence was lower at lower doses. Discontinuation due to adverse events was about 4% across all dose groups.
Less Common but Notable Findings
Gallstones
One Phase 2 participant at 4.5 mg/week developed acute cholelithiasis (gallstones). Gallstone risk is a known class-level signal with rapid weight loss.
Anti-cagrilintide antibodies
46-73% of Phase 2 participants developed anti-cagrilintide antibodies by week 26. Phase 2 datasets did not show a clear effect on weight-loss efficacy or safety.
Cardiovascular safety
A thorough QT study found no clinically relevant QTc prolongation, even at supratherapeutic doses. Long-term cardiovascular outcomes are still being evaluated in REDEFINE 3.
Injection-site reactions
Mild redness or swelling at the injection site is common, especially during early titration. Rotating sites each week reduces local irritation.
Theoretical and Class-Level Risks
These have not been confirmed in cagrilintide trials but are flagged because they appear in related amylin or GLP-1 class data: pancreatitis, gallbladder disease, severe hypoglycemia (mainly when combined with insulin or sulfonylureas), and acute kidney injury secondary to dehydration from severe nausea or vomiting.
Cagrilintide Timeline & What to Monitor
Trial endpoints help set realistic expectations for what cagrilintide does and when. Most published outcomes are measured at 26 weeks (Phase 2) or 68 weeks (Phase 3 REDEFINE).
Trial-Reported Endpoints by Phase
Trial
Lancet 2021 Phase 2
Duration
26 weeks
Population
Overweight/obese, no T2D (n=706)
Mean Weight Loss
10.8% at 4.5 mg vs 3.0% placebo
Trial
Lancet 2023 Phase 2
Duration
32 weeks
Population
T2D + obesity (n=92)
Mean Weight Loss
8.1% cagrilintide alone; 15.6% with semaglutide
Trial
REDEFINE 1 (NEJM 2025)
Duration
68 weeks
Population
Obesity, no T2D (n=3,417)
Mean Weight Loss
11.8% cagrilintide alone; 20.4% CagriSema
Trial
REDEFINE 2 (NEJM 2025)
Duration
68 weeks
Population
T2D + BMI ≥27 (n=1,206)
Mean Weight Loss
13.7% CagriSema vs 3.4% placebo; HbA1c down 1.91 pp
| Trial | Duration | Population | Mean Weight Loss |
|---|---|---|---|
| Lancet 2021 Phase 2 | 26 weeks | Overweight/obese, no T2D (n=706) | 10.8% at 4.5 mg vs 3.0% placebo |
| Lancet 2023 Phase 2 | 32 weeks | T2D + obesity (n=92) | 8.1% cagrilintide alone; 15.6% with semaglutide |
| REDEFINE 1 (NEJM 2025) | 68 weeks | Obesity, no T2D (n=3,417) | 11.8% cagrilintide alone; 20.4% CagriSema |
| REDEFINE 2 (NEJM 2025) | 68 weeks | T2D + BMI ≥27 (n=1,206) | 13.7% CagriSema vs 3.4% placebo; HbA1c down 1.91 pp |
Most trials report mean differences. Individual response varies widely.
What to Monitor During a Cycle
GI tolerance
Nausea, vomiting, constipation, diarrhea — most common during weeks 1-12 of escalation.
Body weight
Trial endpoints used weekly or monthly weights with a standardized scale.
Blood pressure
Both REDEFINE 1 and 2 reported reductions in systolic blood pressure.
Glycemic markers (if T2D)
Fasting glucose and HbA1c improved meaningfully in CagriSema arms; isolated cagrilintide had a smaller effect.
Gallbladder symptoms
Right upper-quadrant pain, especially after rapid weight loss, warrants clinical evaluation.
Injection-site condition
Lipodystrophy or persistent redness suggests rotating sites more aggressively.
Cagrilintide Clinical Evidence Context
Cagrilintide has unusually strong human evidence for a research-use peptide. The development program runs from Phase 1b through three completed Phase 3 readouts, with several more ongoing.
REDEFINE 1 (Phase 3a, NEJM 2025)
Garvey et al., n=3,417, 68 weeks. Cagrilintide monotherapy: 11.8% mean weight loss vs 2.3% placebo. CagriSema: 20.4% vs semaglutide 14.9%. Trial-product estimand.
REDEFINE 2 (Phase 3a, NEJM 2025)
Davies et al., n=1,206, 68 weeks, T2D + BMI ≥27. CagriSema: 13.7% body-weight reduction and 1.91 percentage-point HbA1c reduction vs placebo.
REDEFINE 4 (Phase 3, Feb 2026 readout)
Open-label vs tirzepatide. CagriSema: 23.0% at 84 weeks vs tirzepatide 15 mg: 25.5%. Noninferiority endpoint not met.
Lancet 2021 Phase 2 (monotherapy)
Lau et al., n=706, 26 weeks. Cagrilintide 0.3-4.5 mg vs liraglutide 3 mg/day. 4.5 mg arm: 10.8% mean loss; 2.4 mg arm: 9.7%; liraglutide: 9.0%; placebo: 3.0%.
Lancet 2021 Phase 1b (cagri + sema)
Enebo et al., n=96, 20 weeks. Established additive effect of cagrilintide + semaglutide vs semaglutide alone (17.1% vs 9.8%).
Lancet 2023 Phase 2 (T2D)
Frias et al., n=92, 32 weeks. CagriSema: 15.6% weight loss and 2.2 pp HbA1c reduction. Cagrilintide alone: 8.1%. Semaglutide alone: 5.1%.
Mechanism and pharmacology
Kruse et al. (J Med Chem 2021) characterized the C-20 fatty diacid spacer that extends the half-life. Fletcher et al. (Nature Communications 2025) mapped cagrilintide binding to amylin and calcitonin receptors structurally.
Cardiovascular signal
Gabe et al. (Diabetes Obes Metab 2024) found no clinically relevant QTc prolongation. REDEFINE 3 cardiovascular outcomes trial is ongoing (~7,000 participants).
Evidence Gap
Long-term (multi-year) outcomes data is still maturing. The cardiovascular outcomes endpoint (REDEFINE 3) is event-driven and has not yet reported. Direct head-to-head data against tirzepatide showed CagriSema underperforming the highest tirzepatide dose at 84 weeks — meaning monotherapy claims of comparable efficacy are not supported by current Phase 3 evidence.
Cagrilintide Storage & Handling
Cagrilintide Storage Reference
State
Lyophilized (powder)
Storage
-4F (-20C) freezer
Duration
Up to 24 months
State
Lyophilized (short-term)
Storage
35.6-46.4F (2-8C) refrigerator
Duration
Acceptable for shipping or short storage
State
Reconstituted (liquid)
Storage
35.6-46.4F (2-8C) refrigerator
Duration
28-30 days; do not freeze
| State | Storage | Duration |
|---|---|---|
| Lyophilized (powder) | -4F (-20C) freezer | Up to 24 months |
| Lyophilized (short-term) | 35.6-46.4F (2-8C) refrigerator | Acceptable for shipping or short storage |
| Reconstituted (liquid) | 35.6-46.4F (2-8C) refrigerator | 28-30 days; do not freeze |
Use the supplier's stability data if it is more conservative than the values above.
Cagrilintide shows the same clumping sensitivity seen with many amylin-class peptides. Avoid vigorous shaking during reconstitution, minimize moisture exposure of the lyophilized vial, protect the vial from light, and avoid repeated freeze-thaw cycles. Discard the vial if the solution becomes cloudy, discolored, or develops visible particles.
Cagrilintide Protocol Mistakes & Troubleshooting
Most cagrilintide issues are practical: missed doses, unclear vials, or GI side effects during titration. Below is a short list of the most common scenarios.
- 01
Missed dose, more than 3 days before next scheduled dose
Take the missed dose as soon as possible, then resume the regular weekly schedule. Cagrilintide's 7- to 8-day half-life keeps blood levels relatively stable.
- 02
Missed dose, fewer than 3 days before next scheduled dose
Skip the missed dose and resume the regular schedule. Do not double-dose to make up for it.
- 03
Cloudy or discolored vial after reconstitution
Discard the vial. Cagrilintide should be a clear, colorless solution. Cloudiness suggests aggregation or contamination.
- 04
Wrong BAC volume added
Recalculate concentration with the actual volume used. The amount of cagrilintide is unchanged; what changes is the draw volume per target dose.
- 05
Severe nausea or vomiting during escalation
The published trials commonly held the current dose for an additional 4 weeks before stepping up. Do not skip titration steps. If severe symptoms persist or worsen, seek qualified medical care.
- 06
Persistent injection-site redness or hardness
Rotate injection sites more aggressively (abdomen, front of thigh, upper arm) and avoid reusing the same spot for at least 7 days. Persistent reactions warrant clinical review.
- 07
Right upper-quadrant abdominal pain
Gallstone risk increases with rapid weight loss. Persistent pain in the right upper abdomen requires medical evaluation, not protocol adjustment.
Cagrilintide Regulatory Status
As of May 2026, cagrilintide is investigational and is not FDA-approved in any form. Novo Nordisk filed a New Drug Application for the CagriSema combination (cagrilintide 2.4 mg + semaglutide 2.4 mg) in December 2025, with an FDA decision anticipated in late 2026. Cagrilintide as a standalone monotherapy is being evaluated in the RENEW Phase 3 program; no NDA has been filed for monotherapy at this time.
Internationally, similar regulatory timelines apply. The European Medicines Agency has not approved cagrilintide. Some Phase 3 trial sites are in Asia (REDEFINE 5, REDEFINE 6) but no regulatory approvals have been announced as of May 2026.
Research-use status: Cagrilintide is sold by research peptide suppliers as a lyophilized powder for in-vitro and laboratory research only. It is not approved for human use, compounding through 503A/503B pharmacies, or any clinical purpose outside of registered trials.
Status changes quickly
Regulatory status can change at any time. Verify the current FDA approval status before any research planning by checking the FDA website and ClinicalTrials.gov for the most recent CagriSema and cagrilintide entries.
Cagrilintide vs Semaglutide vs Tirzepatide vs Retatrutide
These compounds all reduce body weight, but they target different receptor systems and produce different magnitudes of effect. They are not interchangeable.
Side-by-Side Comparison
Parameter
Receptors
Cagrilintide
AMY1R/2R/3R + CTR
Semaglutide
GLP-1R
Tirzepatide
GLP-1R + GIPR
Retatrutide
GLP-1R + GIPR + Glucagon-R
Parameter
Half-life
Cagrilintide
~7-8 days
Semaglutide
~7 days
Tirzepatide
~5 days
Retatrutide
~6 days
Parameter
Dosing frequency
Cagrilintide
Once weekly
Semaglutide
Once weekly
Tirzepatide
Once weekly
Retatrutide
Once weekly
Parameter
Max studied dose
Cagrilintide
4.5 mg/week (mono)
Semaglutide
2.4 mg/week
Tirzepatide
15 mg/week
Retatrutide
12 mg/week
Parameter
Peak monotherapy weight loss
Cagrilintide
11.8% at 68 wk
Semaglutide
14.9-17% at 68 wk
Tirzepatide
22.5% at 72 wk
Retatrutide
~24% at 48 wk
Parameter
FDA status (May 2026)
Cagrilintide
Investigational
Semaglutide
Approved (2021)
Tirzepatide
Approved (2023)
Retatrutide
Investigational
Parameter
HbA1c reduction (T2D)
Cagrilintide
~0.9 pp mono; 1.91 pp CagriSema
Semaglutide
1.5-1.8 pp
Tirzepatide
2.0-2.4 pp
Retatrutide
1.6-2.0 pp
Parameter
Notable differentiator
Cagrilintide
Amylin pathway; additive with GLP-1
Semaglutide
Established CV outcomes
Tirzepatide
Highest single-agent obesity efficacy
Retatrutide
Triple-agonist mechanism
| Parameter | Cagrilintide | Semaglutide | Tirzepatide | Retatrutide |
|---|---|---|---|---|
| Receptors | AMY1R/2R/3R + CTR | GLP-1R | GLP-1R + GIPR | GLP-1R + GIPR + Glucagon-R |
| Half-life | ~7-8 days | ~7 days | ~5 days | ~6 days |
| Dosing frequency | Once weekly | Once weekly | Once weekly | Once weekly |
| Max studied dose | 4.5 mg/week (mono) | 2.4 mg/week | 15 mg/week | 12 mg/week |
| Peak monotherapy weight loss | 11.8% at 68 wk | 14.9-17% at 68 wk | 22.5% at 72 wk | ~24% at 48 wk |
| FDA status (May 2026) | Investigational | Approved (2021) | Approved (2023) | Investigational |
| HbA1c reduction (T2D) | ~0.9 pp mono; 1.91 pp CagriSema | 1.5-1.8 pp | 2.0-2.4 pp | 1.6-2.0 pp |
| Notable differentiator | Amylin pathway; additive with GLP-1 | Established CV outcomes | Highest single-agent obesity efficacy | Triple-agonist mechanism |
Numbers reflect published Phase 3 endpoints where available. CagriSema (cagrilintide + semaglutide) reached 20.4% in REDEFINE 1 and 23.0% in REDEFINE 4.
Cagrilintide alone trails semaglutide and tirzepatide in weight-loss magnitude. Its strongest practical use case is combination with semaglutide as CagriSema, where the amylin pathway adds to GLP-1 effects and the combined regimen reached 20.4% weight loss in REDEFINE 1. However, REDEFINE 4 (Feb 2026) found CagriSema did not meet noninferiority versus tirzepatide 15 mg at 84 weeks.
For non-peptide oral options, see the tesofensine protocol, a centrally acting monoamine reuptake inhibitor used in some research-context fat-loss protocols. For the GLP-1 comparison head-to-heads, see the semaglutide protocol, tirzepatide protocol, and retatrutide protocol.
Cagrilintide Blood Tests & Monitoring
Cagrilintide is an amylin-analog research compound usually discussed in weight-loss and metabolic contexts. Monitoring focuses on glucose trends, kidney/liver context, lipids, and symptoms tied to reduced intake or GI effects.
Blood test markers to discuss with a clinician
Marker
A1c
Why it matters
Shows the longer-term blood sugar trend before interpreting weight-loss or appetite changes.
Timing
Baseline
Marker
Fasting glucose
Why it matters
Helps review current glucose control, especially when appetite or food intake changes.
Timing
Follow-up
Marker
Comprehensive metabolic panel (CMP)
Why it matters
Reviews kidney function, liver enzymes, electrolytes, and glucose, which matter when GI symptoms or dehydration occur.
Timing
Baseline
Marker
Lipid panel
Why it matters
Tracks cholesterol and triglyceride changes during weight-loss phases.
Timing
Follow-up
Marker
Blood pressure and resting heart rate
Why it matters
Adds cardiovascular context during body-weight and appetite changes.
Timing
Optional
| Marker | Why it matters | Timing |
|---|---|---|
| A1c | Shows the longer-term blood sugar trend before interpreting weight-loss or appetite changes. | Baseline |
| Fasting glucose | Helps review current glucose control, especially when appetite or food intake changes. | Follow-up |
| Comprehensive metabolic panel (CMP) | Reviews kidney function, liver enzymes, electrolytes, and glucose, which matter when GI symptoms or dehydration occur. | Baseline |
| Lipid panel | Tracks cholesterol and triglyceride changes during weight-loss phases. | Follow-up |
| Blood pressure and resting heart rate | Adds cardiovascular context during body-weight and appetite changes. | Optional |
Monitoring guidance is trial-informed and pathway-based because cagrilintide is not an approved stand-alone medication with a mature clinical monitoring label.
At-home blood test option
Easy at home option to monitor core metrics during research cycles.

Partner link: PDP may earn a commission at no cost to you.
Simple timing framework
Baseline
Discuss baseline labs before starting or escalating, especially with diabetes, kidney disease, gallbladder history, eating-disorder history, or major calorie restriction.
Follow-up
Repeat metabolic markers after 8-12 weeks or sooner if appetite, hydration, vomiting, or glucose symptoms change.
Longer term
For longer protocols, review metabolic and kidney trends every 3-6 months with a clinician.
How to interpret the labs
- GI symptoms and lower intake can affect hydration, electrolytes, and medication needs.
- Diabetes medications may need clinician review if glucose levels change with reduced intake.
- Routine labs do not replace review of abdominal pain, vomiting, gallbladder symptoms, or nutrition status.
Do not wait for routine labs
Severe or persistent abdominal pain, especially with vomiting, needs medical review. Signs of dehydration, fainting, confusion, or inability to keep fluids down should not wait for routine labs.
FAQ
Q1: What is the starting dose of cagrilintide?
It depends on the protocol format. CagriSema-style escalation starts at 0.25 mg per week with semaglutide and steps up every 4 weeks (0.5, 1.0, 1.7, 2.4 mg) over 16 weeks. Monotherapy protocols are commonly initiated at 0.6 mg per week and escalated to a maximum studied dose of 4.5 mg as tolerated.
Q2: What is cagrilintide's half-life?
Plasma half-life is about 159-195 hours, or roughly 6.6-8.1 days. Around the 2.4 mg dose, values near 184 hours have been reported in program summaries. This long half-life is what makes once-weekly dosing possible.
Q3: How much weight loss is reported with cagrilintide?
In REDEFINE 1 (NEJM 2025, n=3,417, 68 weeks), cagrilintide monotherapy at 2.4 mg produced 11.8% mean body-weight reduction versus 2.3% with placebo. CagriSema (cagrilintide + semaglutide) reached 20.4% in the same trial. In Phase 2, monotherapy at 4.5 mg/week reached 10.8% at 26 weeks. Individual response varies.
Q4: How do you reconstitute cagrilintide?
Reconstitute lyophilized cagrilintide with bacteriostatic water by injecting slowly down the inside wall of the vial, then gently swirl until the solution is clear. Do not shake. Common setups are a 5 mg vial with 2.0 mL BAC water (2.5 mg/mL) or a 10 mg vial with 3.0 mL (3.33 mg/mL). For exact unit conversion, use the PepPal calculator.
Q5: Is cagrilintide FDA-approved?
No. As of May 2026, cagrilintide is not FDA-approved in any form. Novo Nordisk filed an NDA for the CagriSema combination in December 2025, with an FDA decision anticipated in late 2026. Standalone cagrilintide (RENEW program) has no NDA filing yet.
Q6: What are the most common cagrilintide side effects?
The most common effects are gastrointestinal and dose-dependent. In the Lancet 2021 Phase 2 trial at 4.5 mg/week, nausea occurred in 47% of participants, injection-site reactions in 43%, constipation in 21%, and vomiting in 8%. Most events were mild to moderate and improved as the dose stabilized.
Q7: How does cagrilintide compare to semaglutide and tirzepatide?
Cagrilintide acts on amylin and calcitonin receptors; semaglutide acts on GLP-1; tirzepatide acts on GLP-1 and GIP. Monotherapy weight-loss magnitude follows tirzepatide (22.5%) > semaglutide (~17%) > cagrilintide (11.8%). Cagrilintide's strongest use is in combination — CagriSema (with semaglutide) reached 20.4% in REDEFINE 1, but did not reach noninferiority versus tirzepatide 15 mg in REDEFINE 4.
Q8: What vial sizes are available for cagrilintide?
Most research suppliers list 5 mg and 10 mg lyophilized vials. At 2.4 mg/week, a 5 mg vial covers about 2 weekly doses and a 10 mg vial covers about 4, before accounting for priming losses.
Q9: How much bacteriostatic water should be added to a cagrilintide vial?
Volume depends on target concentration. Common reconstitutions are 5 mg + 2.0 mL (2.5 mg/mL), 10 mg + 3.0 mL (3.33 mg/mL), or 10 mg + 2.0 mL (5.0 mg/mL). Use the PepPal calculator for exact unit conversions at any concentration.
Q10: What is the maximum cagrilintide dose studied?
The highest monotherapy dose studied is 4.5 mg per week (Lancet 2021 Phase 2). The Phase 3 CagriSema program (REDEFINE 1, 2, REIMAGINE 2) capped cagrilintide at 2.4 mg per week. REDEFINE 11, initiated June 2025, is exploring extended duration and re-escalation strategies.
Q11: How should reconstituted cagrilintide be stored?
Refrigerate at 2-8 degrees Celsius (35.6-46.4 degrees Fahrenheit), protected from light, and use within 28-30 days. Do not freeze the reconstituted solution. Discard the vial if it becomes cloudy, discolored, or develops particles.
Q12: What is the missed-dose rule for cagrilintide?
Because cagrilintide has a 7- to 8-day half-life, the published label-style guidance is to take a missed dose as soon as possible if the next scheduled dose is more than 3 days away, or to skip it if the next dose is closer than 3 days. Do not double-dose.
Q13: What is the current cagrilintide clinical trial program?
REDEFINE (obesity) and REIMAGINE (T2D) are the main Phase 3 programs, with REDEFINE 1, 2, and 4 already reported. REDEFINE 3 evaluates cardiovascular outcomes (~7,000 participants, event-driven). REDEFINE 11, initiated June 2025, tests extended treatment duration. RENEW is the planned monotherapy Phase 3 program.
Q14: Where can I calculate reconstitution and syringe units?
Use the PepPal cagrilintide calculator for any vial size, BAC volume, or target dose conversion to U-100 syringe units.
Q15: Where can I compare research peptide suppliers?
Browse the PepPal supplier directory for current supplier listings, COA verification status, and discount codes.
Sources & Research
- 1. Garvey WT, Blüher M, Osorto Contreras CK, et al. Coadministered Cagrilintide and Semaglutide in Adults with Overweight or Obesity (REDEFINE 1). New England Journal of Medicine (2025)
- 2. Davies MJ, Bajaj HS, Broholm C, et al. Cagrilintide-Semaglutide in Adults with Overweight or Obesity and Type 2 Diabetes (REDEFINE 2). New England Journal of Medicine (2025)
- 3. Lau DCW, Erichsen L, Francisco AM, et al. Once-weekly cagrilintide for weight management in people with overweight and obesity: a Phase 2 trial. The Lancet (2021)
- 4. Enebo LB, Berthelsen KK, Kankam M, et al. Safety, tolerability, pharmacokinetics, and pharmacodynamics of cagrilintide with semaglutide for weight management: a Phase 1b trial. The Lancet (2021)
- 5. Frias JP, Deenadayalan S, Erichsen L, et al. Efficacy and safety of co-administered once-weekly cagrilintide 2.4 mg with semaglutide 2.4 mg in type 2 diabetes: a Phase 2 trial. The Lancet (2023)
- 6. Kruse T, Hansen JL, Dahl K, et al. Development of Cagrilintide, a Long-Acting Amylin Analogue. Journal of Medicinal Chemistry (2021)
- 7. Fletcher MM, Belousoff MJ, Harikumar KG, et al. Structural and dynamic features of cagrilintide binding to calcitonin and amylin receptors. Nature Communications (2025)
- 8. Dehestani B, Stratford NR, le Roux CW. Amylin as a Future Obesity Treatment. Journal of Obesity & Metabolic Syndrome (2021)
- 9. Dutta D, Nagendra L, Harish BG, et al. Efficacy and Safety of Cagrilintide Alone and in Combination with Semaglutide (CagriSema) as Anti-Obesity Medications: A Systematic Review and Meta-Analysis. Indian Journal of Endocrinology and Metabolism (2024)
- 10. D'Ascanio AM, Mullally JA, Frishman WH. Cagrilintide: A Long-Acting Amylin Analog for the Treatment of Obesity. Cardiology in Review (2024)
- 11. Novo Nordisk (press release). CagriSema 2.4 mg / 2.4 mg demonstrated 22.7% mean weight reduction in adults with overweight or obesity in REDEFINE 1, published in NEJM. PR Newswire (2025)
- 12. Novo Nordisk (press release). Novo Nordisk files for FDA approval of CagriSema (December 18, 2025). PR Newswire (2025)
- 13. ClinicalTrials.gov. REDEFINE 1 (NCT05567796), REDEFINE 2 (NCT05394519), REDEFINE 3 (NCT05669755), REDEFINE 4 (NCT06131437), REDEFINE 11 (NCT07011667), REIMAGINE 2 (NCT06065540). U.S. National Library of Medicine (2026)
Related Dosing Protocols
Written by Garret Grant
Founder & Lead Researcher · B.S. Civil Engineering, UCLA
Last updated: May 2026
Human-researched and AI-assisted with full editorial review. I verify sources, protocol interpretation, and final judgments personally. See methodology.
Share this page

