Melanotan 2 Quick Start
Melanotan 2 (also written MT-2, MT-II, or melanotan ii) is a lab-made peptide. It was first developed at the University of Arizona in the 1980s and 1990s. The goal was to find a way to trigger tanning without long sun exposure.
MT-2 copies a natural hormone in the body called alpha-MSH. Alpha-MSH helps control skin color. The lab version is stronger and lasts longer than the natural one, which is why it has bigger effects but also more side effects.
Most research workflows use a daily loading phase followed by a weekly maintenance dose. The most common vial size is 10 mg, and the most common route is subcutaneous (SubQ) injection. Some studies also tested intranasal use.
Use this page for dose steps, timing, vial mixing, and storage. Want the larger research view? See the Melanotan II safety guide on Peptide Advisors.
Route
Subcutaneous (SubQ) injection is the most studied route. Intranasal spray has been tested but is less predictable.
Measure
Use a U-100 insulin syringe. The unit math depends on how much BAC water you mix with the 10 mg vial.
Loading
A common research framework starts at 100-250 mcg daily and escalates to 500-1000 mcg as nausea tolerance improves.
Maintenance
After loading, many users drop to 1-2 doses per week to hold pigmentation.
Research status
Not FDA-approved. Illegal to sell for human use in the US, UK, and Australia.
Disclaimer
This page is an educational research reference. It is not medical advice and not a treatment plan. Melanotan 2 is not FDA-approved and is sold for research use only.
Need exact syringe units? Open the Pep Pal calculator and enter your vial size and BAC water volume.
Melanotan 2 Dosing Protocol & Schedule
There is no FDA-approved dose for Melanotan 2. The schedules below are common research-planning frameworks built from early human studies and community use. They are not personal medical advice.
Most workflows use two phases. Loading is daily dosing to build pigment. Maintenance is one or two doses per week to hold it. Nausea is the main reason people slow down or pause dose increases.
Melanotan 2 Dosing Framework
Pick the phase you are researching to see the typical dose and timing notes.
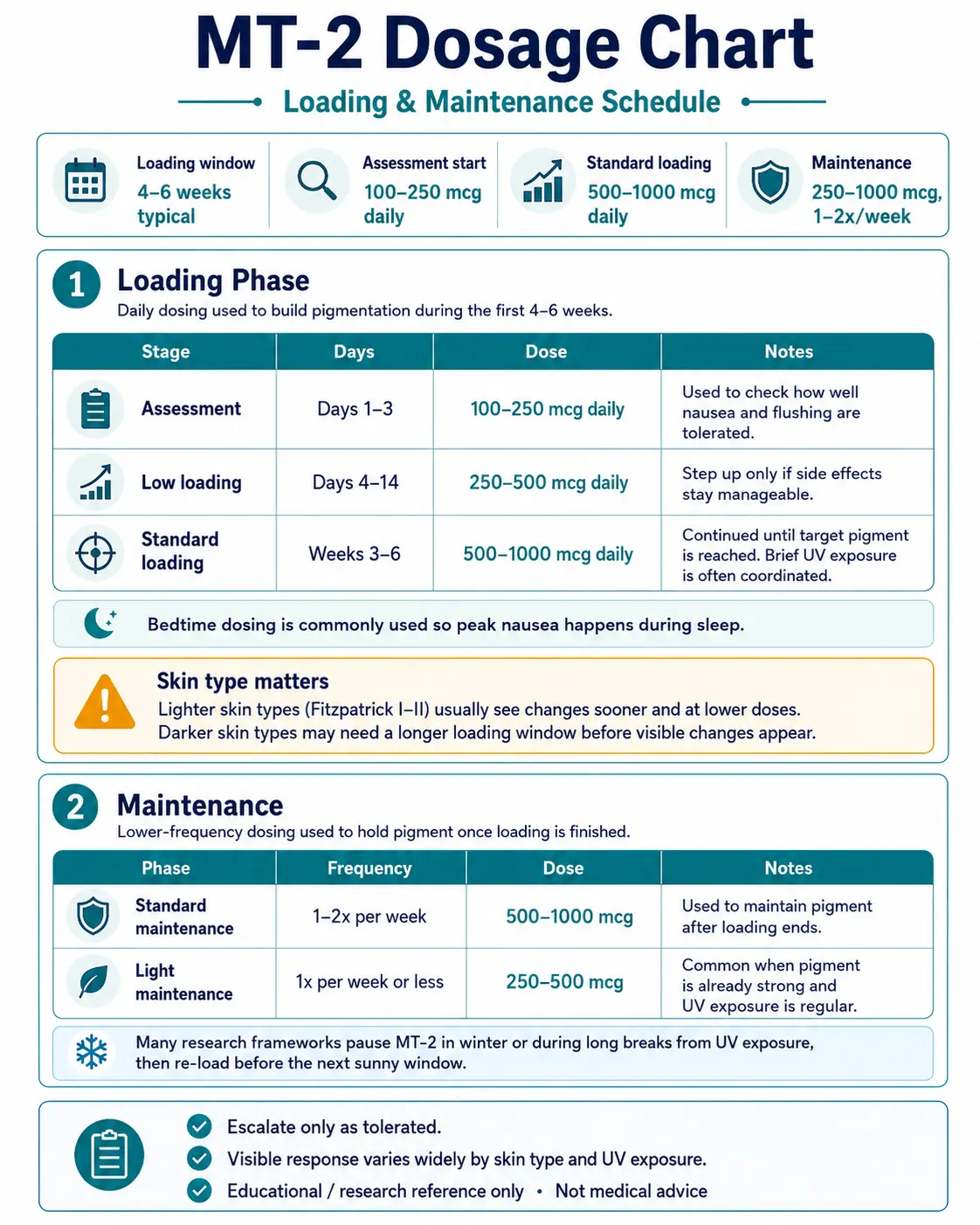
Daily dosing used to build pigmentation during the first 4-6 weeks.
Loading Phase - Daily Dosing
Stage
Assessment
Days
Days 1-3
Dose
100-250 mcg daily
Notes
Used to check how well nausea and flushing are tolerated.
Stage
Low loading
Days
Days 4-14
Dose
250-500 mcg daily
Notes
Step up only if side effects stay manageable.
Stage
Standard loading
Days
Weeks 3-6
Dose
500-1000 mcg daily
Notes
Continued until target pigment is reached. Brief UV exposure is often coordinated.
| Stage | Days | Dose | Notes |
|---|---|---|---|
| Assessment | Days 1-3 | 100-250 mcg daily | Used to check how well nausea and flushing are tolerated. |
| Low loading | Days 4-14 | 250-500 mcg daily | Step up only if side effects stay manageable. |
| Standard loading | Weeks 3-6 | 500-1000 mcg daily | Continued until target pigment is reached. Brief UV exposure is often coordinated. |
Bedtime dosing is commonly used so peak nausea happens during sleep.
Skin type matters
Lighter skin types (Fitzpatrick I-II) usually see changes sooner and at lower doses. Darker skin types may need a longer loading window before visible changes appear.
Lower-frequency dosing used to hold pigment once loading is finished.
Maintenance Dosing
Phase
Standard maintenance
Frequency
1-2x per week
Dose
500-1000 mcg
Notes
Used to maintain pigment after loading ends.
Phase
Light maintenance
Frequency
1x per week or less
Dose
250-500 mcg
Notes
Common when pigment is already strong and UV exposure is regular.
| Phase | Frequency | Dose | Notes |
|---|---|---|---|
| Standard maintenance | 1-2x per week | 500-1000 mcg | Used to maintain pigment after loading ends. |
| Light maintenance | 1x per week or less | 250-500 mcg | Common when pigment is already strong and UV exposure is regular. |
Many research frameworks pause MT-2 in winter or during long breaks from UV exposure, then re-load before the next sunny window.
Higher per-dose levels used in early erectile-dysfunction studies. Listed for reference, not as a recommendation.
Trial-Level Dosing Used in ED Research
Context
ED trial dose
Dose
0.025 mg/kg (about 1.75 mg at 70 kg / 154 lb)
Notes
Used in Phase II ED studies. Caused more nausea than typical tanning doses.
Context
Community low-dose context
Dose
500 mcg to 1 mg
Notes
Reported by users seeking sexual-function effects with less GI burden.
| Context | Dose | Notes |
|---|---|---|
| ED trial dose | 0.025 mg/kg (about 1.75 mg at 70 kg / 154 lb) | Used in Phase II ED studies. Caused more nausea than typical tanning doses. |
| Community low-dose context | 500 mcg to 1 mg | Reported by users seeking sexual-function effects with less GI burden. |
These figures describe published study arms and community reports. They are not dosing recommendations.
Cycle Guidelines
Common Cycle Lengths
Approach
Short cycle
Duration
4 weeks
Off Period
4+ weeks
Best For
First-time research planning
Approach
Standard cycle
Duration
6 weeks
Off Period
4-6 weeks
Best For
Building visible pigment
Approach
Extended cycle
Duration
8 weeks
Off Period
6+ weeks
Best For
Slower titration or stronger nausea profile
| Approach | Duration | Off Period | Best For |
|---|---|---|---|
| Short cycle | 4 weeks | 4+ weeks | First-time research planning |
| Standard cycle | 6 weeks | 4-6 weeks | Building visible pigment |
| Extended cycle | 8 weeks | 6+ weeks | Slower titration or stronger nausea profile |
Long-term controlled safety data is limited. Many frameworks cap loading at 8 weeks.
Evidence boundary
No FDA-approved dose exists. Loading and maintenance models are community-derived and informed by small early-phase studies. Personal medical decisions should involve a licensed clinician.
Melanotan 2 Reconstitution Guide
Melanotan 2 ships as a dry white powder in a 10 mg vial. You mix it with bacteriostatic (BAC) water to turn it into a liquid you can draw into a syringe. The amount of water you add changes the concentration and the unit math.
Find the BAC water volume you plan to use in the table below, then read across to see the syringe units for your target dose. All units assume a U-100 (1 mL = 100 units) insulin syringe.
10 mg Vial - Reconstitution Math
BAC Water Added
1.0 mL
Concentration
10 mg/mL
250 mcg
0.025 mL (2.5 u)
500 mcg
0.05 mL (5 u)
750 mcg
0.075 mL (7.5 u)
1 mg
0.10 mL (10 u)
BAC Water Added
2.0 mL
Concentration
5 mg/mL
250 mcg
0.05 mL (5 u)
500 mcg
0.10 mL (10 u)
750 mcg
0.15 mL (15 u)
1 mg
0.20 mL (20 u)
BAC Water Added
3.0 mL
Concentration
3.33 mg/mL
250 mcg
0.075 mL (7.5 u)
500 mcg
0.15 mL (15 u)
750 mcg
0.225 mL (22.5 u)
1 mg
0.30 mL (30 u)
BAC Water Added
5.0 mL
Concentration
2 mg/mL
250 mcg
0.125 mL (12.5 u)
500 mcg
0.25 mL (25 u)
750 mcg
0.375 mL (37.5 u)
1 mg
0.50 mL (50 u)
| BAC Water Added | Concentration | 250 mcg | 500 mcg | 750 mcg | 1 mg |
|---|---|---|---|---|---|
| 1.0 mL | 10 mg/mL | 0.025 mL (2.5 u) | 0.05 mL (5 u) | 0.075 mL (7.5 u) | 0.10 mL (10 u) |
| 2.0 mL | 5 mg/mL | 0.05 mL (5 u) | 0.10 mL (10 u) | 0.15 mL (15 u) | 0.20 mL (20 u) |
| 3.0 mL | 3.33 mg/mL | 0.075 mL (7.5 u) | 0.15 mL (15 u) | 0.225 mL (22.5 u) | 0.30 mL (30 u) |
| 5.0 mL | 2 mg/mL | 0.125 mL (12.5 u) | 0.25 mL (25 u) | 0.375 mL (37.5 u) | 0.50 mL (50 u) |
2 mL is the most common setup because it gives clean unit math at 500 mcg (10 units).
Step-by-Step Reconstitution
- 01
Wipe the stoppers
Use a fresh alcohol swab on the Melanotan 2 vial stopper and the BAC water vial stopper.
- 02
Draw your BAC water
Pull the planned volume (usually 2 mL) into a sterile syringe.
- 03
Inject against the side wall
Aim the needle at the inside wall of the vial, not directly onto the powder. This protects the peptide.
- 04
Let it dissolve
Set the vial down. The powder will dissolve on its own. Gently swirl if needed. Do not shake hard.
- 05
Check the solution
The liquid should look clear and colorless. If it is cloudy or has visible particles, do not use it.
- 06
Label and date
Write the date and final concentration on the vial. This helps track shelf life.
- 07
Refrigerate
Store the reconstituted vial at 35.6-46.4F (2-8C). Keep it out of direct light.
Need exact units for a different vial size?
Use the free Pep Pal calculator to plug in your own vial size, BAC water volume, and target dose.
Melanotan 2 Dosage Chart
This Melanotan 2 dosage chart summarizes the common loading and maintenance framework, with dose escalation shown by phase and schedule.
How Melanotan 2 Works
Melanotan 2 works by turning on a family of receptors in your body called melanocortin receptors. These receptors sit on different cells and control different things: skin pigment, hunger, sexual signaling, and more.
Most modern drugs are designed to hit only one receptor. MT-2 hits several at once. That is why it produces several effects at the same time - and also more side effects than newer, targeted versions.
MC1R - tanning
MC1R sits on the pigment-making cells in your skin. When MT-2 turns it on, those cells make more melanin, which is the natural brown pigment that darkens skin.
MC3R and MC4R - sexual signaling
MC3R and MC4R sit in the brain and control sexual arousal. This is a brain-level effect, not a blood-flow effect like Viagra.
MC4R - appetite
The same brain receptors involved in arousal also help control hunger. Many users report less appetite while loading.
MC5R - secondary effects
MC5R sits mostly in glands. It is less linked to obvious effects but may add to the overall side-effect picture.
Because MT-2 turns on all of these receptors at once, researchers later built more focused versions. Afamelanotide mostly hits MC1R and is FDA-approved for a rare skin condition called erythropoietic protoporphyria. Bremelanotide (PT-141) mostly hits MC3R/MC4R and is FDA-approved for low sexual desire in women.
Melanotan 2 Supplies Needed
The math below assumes a 10 mg vial, 2 mL BAC water, and 500 mcg daily during loading.
Recommended Supply
Use discount code PEPPAL at eligible peptide supplier checkouts.

Melanotan 2 Supply

SiPhox Health At-Home Blood Test
Injection Supplies
Disclosure: supply links may earn PDP a commission at no cost to you.
Peptide Vials
A 10 mg vial provides about 20 doses at 500 mcg per day.
| Cycle length | Planning note |
|---|---|
4 weeks 2 vials | 28 daily doses planned; one vial covers ~20 doses. |
6-8 weeks 3 vials | 6 weeks: 42 daily doses planned; allow margin for waste.; 8 weeks: 56 daily doses planned; some users use 4 if they dose higher. |
4 weeks
2 vials
28 daily doses planned; one vial covers ~20 doses.
6-8 weeks
3 vials
6 weeks: 42 daily doses planned; allow margin for waste.; 8 weeks: 56 daily doses planned; some users use 4 if they dose higher.
Insulin Syringes (U-100)
Use 0.3 mL / 30-unit syringes for cleaner draws at small dose volumes.
| Cycle length | Planning note |
|---|---|
4 weeks 30 syringes | 1 per injection, plus a few spares. |
6 weeks 45 syringes | 1 per injection. |
8 weeks 60 syringes | 1 per injection. |
4 weeks
30 syringes
1 per injection, plus a few spares.
6 weeks
45 syringes
1 per injection.
8 weeks
60 syringes
1 per injection.
Bacteriostatic Water
Plan 2 mL per 10 mg vial. A 10 mL bottle covers several vials.
| Cycle length | Planning note |
|---|---|
4-8 weeks 1 x 10 mL bottle | 4 weeks: Two vials use 4 mL total.; 6 weeks: Three vials use 6 mL total.; 8 weeks: Three vials use 6 mL total; a second bottle adds margin. |
4-8 weeks
1 x 10 mL bottle
4 weeks: Two vials use 4 mL total.; 6 weeks: Three vials use 6 mL total.; 8 weeks: Three vials use 6 mL total; a second bottle adds margin.
Round up for priming losses, dropped syringes, and damaged swabs. Mole-monitoring tools (a phone camera and a notes app) are also worth setting up before starting.
Who Melanotan 2 Is For and Who Should Avoid It
MT-2 is a research compound. It is not approved for any medical use. People typically researching it are interested in tanning response, melanocortin signaling, or appetite/sexual function research. Personal use carries real risk and should involve a clinician.
The groups below have higher risk and are usually flagged in case reports and clinical commentary.
Personal or family history of melanoma
Case reports have linked MT-2 use to mole changes and melanoma diagnoses. People with prior skin cancer or strong family history should avoid it.
Many moles or atypical moles
MT-2 darkens existing moles and can trigger new ones. Anyone with lots of moles or atypical (dysplastic) moles is at higher risk of missing a real warning sign.
Cardiovascular conditions
Flushing, blood pressure changes, and rare reports of more serious events suggest people with uncontrolled cardiovascular disease should avoid use.
History of priapism or sickle cell trait
Case reports include priapism, which is a prolonged, painful erection that needs medical care. Anyone with a higher baseline risk should avoid MT-2.
Pregnancy and breastfeeding
No human safety data exists for these groups. MT-2 should not be used during pregnancy or while breastfeeding.
Active medications that affect blood pressure or arousal
There is little drug-interaction data. People on blood pressure, ED, or psychiatric medications should review options with a clinician first.
Melanotan 2 Side Effects & Safety
Most MT-2 side effects show up early in loading and at higher doses. Many fade as the body adjusts, but a few are more serious and need attention.
Common: nausea and flushing
Nausea is the most common reason people slow down or stop. Flushing (a warm, red feeling in the face and chest) is also frequent. Both tend to ease over the first 1-2 weeks of dosing.
Common: appetite changes, fatigue, yawning
Many users report eating less, feeling sleepy, or yawning soon after a dose. These are linked to the same melanocortin pathways that affect the brain.
Sexual: spontaneous erections and increased desire
Trial data show MT-2 can cause unplanned erections and higher sexual desire. This is a brain-signaling effect, not a blood-flow drug like Viagra.
Dermatologic: darkening moles and new moles
MT-2 can darken existing moles, freckles, and other spots. Case reports describe new moles appearing and rapid changes in old ones after only one or two doses. Because mole changes can sometimes signal melanoma (a serious skin cancer), close monitoring is important.
Serious: priapism
A few case reports describe priapism, which is a painful, lasting erection. It is a medical emergency and needs urgent care.
Serious: rhabdomyolysis at overdose levels
Overdose case reports describe rhabdomyolysis, which is when muscle tissue breaks down and can harm the kidneys. These cases involved doses far above typical research-planning ranges.
Watch your moles closely
Photograph your moles before starting and every few weeks during use. If any mole changes shape, color, size, or starts to bleed, stop dosing and see a dermatologist.
For a broader class-level safety view, see the PepPal peptide side effects guide.
Melanotan 2 Timeline & What to Monitor
Tanning is not instant. The body needs time to make new melanin and move it into the skin. Sexual and appetite effects can show up faster but vary widely from person to person.
What to Expect by Week
Window
Days 1-3
Likely Pattern
Most nausea and flushing happen here. Skin usually looks the same.
What to Track
Tolerance level, whether bedtime dosing helps with nausea.
Window
Weeks 1-2
Likely Pattern
Some users notice freckling or slight darkening, especially with light UV exposure.
What to Track
Mole photos, first signs of pigment change, side-effect intensity.
Window
Weeks 3-4
Likely Pattern
Pigment changes are more visible. Sexual and appetite effects often plateau.
What to Track
Compare baseline mole photos. Note if any mole looks different.
Window
Weeks 5-8
Likely Pattern
Loading often wraps up here. Many users move to weekly maintenance.
What to Track
Decide if the result is enough or if more loading is needed.
Window
After loading
Likely Pattern
Pigment fades slowly without maintenance dosing or regular UV exposure.
What to Track
How long pigment lasts at your chosen maintenance dose.
| Window | Likely Pattern | What to Track |
|---|---|---|
| Days 1-3 | Most nausea and flushing happen here. Skin usually looks the same. | Tolerance level, whether bedtime dosing helps with nausea. |
| Weeks 1-2 | Some users notice freckling or slight darkening, especially with light UV exposure. | Mole photos, first signs of pigment change, side-effect intensity. |
| Weeks 3-4 | Pigment changes are more visible. Sexual and appetite effects often plateau. | Compare baseline mole photos. Note if any mole looks different. |
| Weeks 5-8 | Loading often wraps up here. Many users move to weekly maintenance. | Decide if the result is enough or if more loading is needed. |
| After loading | Pigment fades slowly without maintenance dosing or regular UV exposure. | How long pigment lasts at your chosen maintenance dose. |
Mole photos
Take wide and close-up phone photos of every visible mole before you start. Re-photograph every 2-4 weeks. Compare them side by side.
Skin exam
A baseline full-skin exam with a dermatologist before starting is the most useful single safety step, especially for anyone with many moles or fair skin.
Blood pressure
If you have any blood pressure history, check it before and during use. Stop if you see a clear unexplained rise.
Side-effect log
Note nausea level, flushing, sleep changes, and any new symptoms. This helps you decide when to escalate or pause.
Melanotan 2 Clinical Evidence Context
The published human evidence for MT-2 is small and old. Most trials were done in the 1990s and early 2000s with 20 to 100 participants. They showed real tanning and sexual-function effects but also high nausea rates at clinical doses. No large modern trials have been done.
Dorr et al. 1996 - Phase I, healthy volunteers
Showed clear pigmentation gains with frequent nausea and spontaneous erections at studied doses.
Wessells et al. 1998 - Phase II, psychogenic ED
Placebo-controlled study reporting strong erection-response signals in most treated participants.
Wessells et al. 2000 (Urology) - Phase II, organic ED
Reported better erection response and rigidity time vs. placebo in men with organic ED.
Wessells et al. 2000 (combined) - mixed ED
Combined analysis showing higher sexual desire and robust erection frequency, with notable nausea rates.
Dorr et al. 2004 - Phase I, MT-2 plus UV
MT-2 combined with brief UV exposure produced stronger tanning than UV alone in healthy volunteers.
Minakova et al. 2019 - preclinical mouse model
Animal study suggesting MT-2 may affect social behavior through MC4R-oxytocin signaling. Not human evidence.
Bottom line: small early studies confirmed MT-2 produces measurable tanning and sexual-function effects, but at the cost of nausea and a broad side-effect profile. Rather than continue MT-2 development, researchers built more selective successors: afamelanotide for tanning (FDA-approved for a rare skin condition) and bremelanotide / PT-141 for sexual function (FDA-approved for low sexual desire in women). MT-2 itself was never submitted for FDA approval.
Melanotan 2 Storage & Handling
Storage by Form
Form
Lyophilized (powder)
Temperature
-4F (-20C) freezer
Duration
Long-term (years)
Form
Lyophilized (powder)
Temperature
35.6-46.4F (2-8C)
Duration
Several months
Form
Lyophilized (powder)
Temperature
Room temperature
Duration
Shipping window (weeks)
Form
Reconstituted
Temperature
35.6-46.4F (2-8C)
Duration
2-4 weeks
Form
Reconstituted aliquots
Temperature
-4F (-20C)
Duration
3-4 months
| Form | Temperature | Duration |
|---|---|---|
| Lyophilized (powder) | -4F (-20C) freezer | Long-term (years) |
| Lyophilized (powder) | 35.6-46.4F (2-8C) | Several months |
| Lyophilized (powder) | Room temperature | Shipping window (weeks) |
| Reconstituted | 35.6-46.4F (2-8C) | 2-4 weeks |
| Reconstituted aliquots | -4F (-20C) | 3-4 months |
Protect the vial from light, use BAC water (not regular water) for multi-dose handling, and split into single-use aliquots before freezing to avoid repeat thaw cycles.
Melanotan 2 Protocol Mistakes & Troubleshooting
Nausea is too strong
Cut the dose in half and dose at bedtime. If nausea is still hard to manage, pause loading for a few days. Restart slower.
Mole looks different
Stop dosing. Photograph the mole. See a dermatologist before resuming anything.
Missed dose
Skip the missed dose and continue the next day. Pigment builds slowly, so one missed dose has little impact.
Cloudy or off-color vial
Do not use it. Cloudiness suggests bacterial contamination or breakdown. Reconstitute a new vial.
No visible tanning after 3-4 weeks
Check three things: vial concentration, syringe math, and UV exposure. Many users do not see pigment without brief, controlled sunlight or tanning-bed exposure.
Unexpected erection or strong arousal
This is a known central effect. Lower the dose or switch dosing to a time when this is easier to manage.
Storage mistake
If a reconstituted vial was left at room temperature for more than a few hours, do not use it. Refrigerate fresh batches as soon as they are mixed.
Melanotan 2 Regulatory Status
As of May 2026, Melanotan 2 is not approved by the FDA (US), EMA (EU), MHRA (UK), or TGA (Australia) for any indication. It is sold only as a research-use compound. Selling it for human use is illegal in the US, UK, and Australia.
Not FDA-approved
No company has submitted Melanotan 2 for FDA approval. Approved melanocortin drugs are afamelanotide (Scenesse) and bremelanotide (Vyleesi).
Not a supplement
MT-2 is not legal as a dietary supplement. Products labeled as sprays or tanning kits are sold without regulatory approval.
International status
Banned for human-use sale in the UK and Australia. The European Medicines Agency has issued warnings against unregulated use.
Research-use only
Vendors typically label MT-2 as 'for research purposes only / not for human consumption.' This label is a legal boundary, not a quality guarantee.
Melanotan 2 vs PT-141 vs Afamelanotide
MT-2, PT-141 (bremelanotide), and afamelanotide (sometimes called Melanotan I) all started from the same source - the natural hormone alpha-MSH. The key difference is how specific each one is. MT-2 hits many receptors. The other two were built to focus on one job.
Side-by-Side Comparison
Feature
Structure
Melanotan 2
Cyclic 7-amino acid peptide
Afamelanotide (MT-I)
Linear 13-amino acid peptide
PT-141 (Bremelanotide)
Cyclic 7-amino acid derivative
Feature
Receptor focus
Melanotan 2
MC1R + MC3R + MC4R + MC5R (non-selective)
Afamelanotide (MT-I)
MC1R-preferring
PT-141 (Bremelanotide)
MC3R/MC4R-preferring
Feature
Plasma half-life
Melanotan 2
~1-2 hours
Afamelanotide (MT-I)
~0.8-1.7 hours
PT-141 (Bremelanotide)
~2.7 hours
Feature
Main effect
Melanotan 2
Tanning + sexual + appetite changes
Afamelanotide (MT-I)
Tanning and photoprotection
PT-141 (Bremelanotide)
Sexual arousal and desire
Feature
Tanning strength
Melanotan 2
Strong
Afamelanotide (MT-I)
Strong
PT-141 (Bremelanotide)
Minimal
Feature
Sexual effect
Melanotan 2
Strong in ED studies
Afamelanotide (MT-I)
Not primary
PT-141 (Bremelanotide)
Primary use case
Feature
Dosing
Melanotan 2
Daily loading, then 1-2x weekly
Afamelanotide (MT-I)
Subcutaneous implant
PT-141 (Bremelanotide)
On-demand, max 1 per 24 hours
Feature
FDA status
Melanotan 2
Not approved
Afamelanotide (MT-I)
Approved (Scenesse) for EPP
PT-141 (Bremelanotide)
Approved (Vyleesi) for low female sexual desire
Feature
Nausea burden
Melanotan 2
High and dose-limiting
Afamelanotide (MT-I)
Lower in selective use
PT-141 (Bremelanotide)
Common, listed on label
| Feature | Melanotan 2 | Afamelanotide (MT-I) | PT-141 (Bremelanotide) |
|---|---|---|---|
| Structure | Cyclic 7-amino acid peptide | Linear 13-amino acid peptide | Cyclic 7-amino acid derivative |
| Receptor focus | MC1R + MC3R + MC4R + MC5R (non-selective) | MC1R-preferring | MC3R/MC4R-preferring |
| Plasma half-life | ~1-2 hours | ~0.8-1.7 hours | ~2.7 hours |
| Main effect | Tanning + sexual + appetite changes | Tanning and photoprotection | Sexual arousal and desire |
| Tanning strength | Strong | Strong | Minimal |
| Sexual effect | Strong in ED studies | Not primary | Primary use case |
| Dosing | Daily loading, then 1-2x weekly | Subcutaneous implant | On-demand, max 1 per 24 hours |
| FDA status | Not approved | Approved (Scenesse) for EPP | Approved (Vyleesi) for low female sexual desire |
| Nausea burden | High and dose-limiting | Lower in selective use | Common, listed on label |
These three compounds share melanocortin ancestry, but they are not interchangeable. Receptor focus and regulatory status make them different tools.
For deeper compound-specific guides, see the PT-141 protocol and the afamelanotide protocol.
Melanotan II Blood Tests & Monitoring
Melanotan II is usually discussed in pigment and melanocortin-pathway research. Monitoring is less about routine labs and more about skin history, blood pressure context, and symptom review.
Blood test markers to discuss with a clinician
Marker
Blood pressure and resting heart rate
Why it matters
Adds cardiovascular context because melanocortin-pathway compounds may affect blood pressure, flushing, or nausea symptoms.
Timing
Baseline
Marker
Comprehensive metabolic panel (CMP)
Why it matters
Provides broad liver, kidney, electrolyte, and glucose context before systemic exposure.
Timing
Baseline
Marker
CBC with differential
Why it matters
Adds a broad health screen before interpreting systemic symptoms.
Timing
Optional
Marker
Pregnancy test
Why it matters
Pregnancy status matters before exposure in people who can become pregnant.
Timing
Baseline
| Marker | Why it matters | Timing |
|---|---|---|
| Blood pressure and resting heart rate | Adds cardiovascular context because melanocortin-pathway compounds may affect blood pressure, flushing, or nausea symptoms. | Baseline |
| Comprehensive metabolic panel (CMP) | Provides broad liver, kidney, electrolyte, and glucose context before systemic exposure. | Baseline |
| CBC with differential | Adds a broad health screen before interpreting systemic symptoms. | Optional |
| Pregnancy test | Pregnancy status matters before exposure in people who can become pregnant. | Baseline |
Monitoring guidance is melanocortin-pathway-based, with emphasis on skin surveillance and cardiovascular context.
At-home blood test option
Easy at home option to monitor core metrics during research cycles.

Partner link: PDP may earn a commission at no cost to you.
Simple timing framework
Baseline
Discuss baseline review before starting, especially with personal or family skin-cancer history, changing moles, high blood pressure, pregnancy potential, or sexual-function medication use.
Follow-up
Review skin changes, blood pressure, and symptoms after early exposure or dose changes.
Longer term
For repeated exposure, periodic skin checks with a clinician or dermatologist may be more useful than broad lab repetition.
How to interpret the labs
- Mole changes and skin history matter more than routine bloodwork for pigment-related concerns.
- Dermatology review is especially relevant with changing lesions, many moles, or skin-cancer history.
- Nausea, flushing, libido changes, and blood pressure symptoms should be interpreted with medication context.
Do not wait for routine labs
Changing moles, bleeding lesions, chest pain, fainting, severe headache, or allergic symptoms need medical review.
FAQ
Q1: What is the starting dose of Melanotan 2?
Many research frameworks start between 100 and 250 mcg daily for the first 3 days. This is used to check how well nausea and flushing are tolerated. Common loading then steps up to 500 mcg daily.
Q2: What is Melanotan 2's half-life?
The plasma half-life is about 1-2 hours, meaning MT-2 clears the bloodstream that quickly. The effects (tanning, appetite, sexual signaling) can last 24-48+ hours because they depend on slower biological changes. Online claims of a '33-hour half-life' confuse those two things.
Q3: How fast can someone expect tanning results?
Most users see some pigment change in 1-2 weeks during loading. Stronger tanning often develops over 4-8 weeks. Lighter skin types usually see changes faster than darker skin types.
Q4: How do you reconstitute Melanotan 2?
A common setup is a 10 mg vial mixed with 2 mL of bacteriostatic water. That gives 5 mg/mL. At that concentration, a 500 mcg dose equals 0.10 mL or 10 units on a U-100 insulin syringe. For other vial or water volumes, use the PepPal calculator.
Q5: Is Melanotan 2 FDA-approved?
No. As of May 2026, MT-2 is not approved by the FDA, EMA, MHRA, or TGA for any use. It is sold only as a research compound and is illegal to sell for human use in the US, UK, and Australia.
Q6: What are the most common Melanotan 2 side effects?
Nausea and flushing are the most common, especially in the first 1-2 weeks. Appetite drop, fatigue, and yawning are also frequent. Mole darkening and new moles can occur and need to be monitored.
Q7: Is Melanotan 2 linked to skin cancer?
Case reports have documented mole darkening, new moles, and rare melanoma diagnoses after MT-2 use. A direct cause has not been proven in a large trial, but most dermatologists recommend a baseline skin exam, regular mole photography, and stopping use if any mole changes shape, color, or size.
Q8: How does Melanotan 2 compare to PT-141?
MT-2 is non-selective and produces tanning, sexual, and appetite effects. PT-141 (bremelanotide) is more selective for MC3R/MC4R and is FDA-approved for low sexual desire in women under the brand name Vyleesi. PT-141 has minimal tanning effect.
Q9: How does Melanotan 2 compare to afamelanotide?
Afamelanotide (sometimes called Melanotan I) is MC1R-preferring and is FDA-approved as Scenesse for a rare skin condition called erythropoietic protoporphyria. It is delivered as a subcutaneous implant. MT-2 hits more receptors and has a broader side-effect profile.
Q10: Melanotan 2 nasal spray vs injection - what's the difference?
Subcutaneous injection is the most studied route and has predictable dose delivery. Intranasal spray has been tested in limited research and varies more from dose to dose because absorption through the nasal lining is less consistent. Most published dosing data is from injection.
Q11: What vial size is most common?
10 mg lyophilized vials are the standard format for research-grade Melanotan 2.
Q12: How much BAC water should be added to a 10 mg vial?
2 mL is the most common choice because the unit math at 500 mcg comes out to a clean 10 units. 1 mL, 3 mL, and 5 mL are also used. The reconstitution table above shows the syringe units for each option.
Q13: What's the highest dose studied in trials?
Early clinical studies reached around 0.025-0.03 mg/kg, which is roughly 1.75 mg at 70 kg (154 lb). Overdose case reports describe severe toxicity (including rhabdomyolysis) at much higher self-administered doses.
Q14: How should reconstituted Melanotan 2 be stored?
Store reconstituted vials at 35.6-46.4F (2-8C) and protect from light. For longer storage, split the vial into single-use aliquots and freeze them. Avoid repeat freeze-thaw cycles.
Q15: Why did Melanotan 2 development stop while PT-141 and afamelanotide moved forward?
MT-2's side-effect profile and broad receptor activity made it difficult to develop for any one approved use. Researchers pivoted to more selective compounds: afamelanotide for tanning indications and PT-141 for sexual function. Both reached FDA approval. MT-2 never did.
Q16: Is this medical advice?
No. This page is an educational research reference. Personal medical decisions should involve a licensed clinician. Peptide Dosing Protocols does not sell Melanotan 2 and is not responsible for off-label use.
Sources & Research
- 1. Dorr RT, Lines R, Levine N, et al. Evaluation of Melanotan-II, a superpotent cyclic melanotropic peptide in a pilot phase-I clinical study. Life Sciences (1996)
- 2. Wessells H, Fuciarelli K, Hansen J, et al. Synthetic melanotropic peptide initiates erections in men with psychogenic erectile dysfunction. Journal of Urology (1998)
- 3. Wessells H, Gralnek D, Dorr R, et al. Effect of an alpha-melanocyte stimulating hormone analog on penile erection and sexual desire in men with organic erectile dysfunction. Urology (2000)
- 4. Wessells H, Levine N, Hadley ME, et al. Melanocortin receptor agonists, penile erection, and sexual motivation: human studies with Melanotan II. International Journal of Impotence Research (2000)
- 5. Dorr RT, Ertl GA, Levine N, et al. Effects of a superpotent melanotropic peptide in combination with solar UV radiation on tanning of the skin in human volunteers. Archives of Dermatology (2004)
- 6. Nelson ME, Bryant SM, Aks SE. Melanotan II injection resulting in systemic toxicity and rhabdomyolysis. Clinical Toxicology (2012)
- 7. Hjuler KF, Lorentzen HF. Melanoma associated with the use of melanotan-II. Dermatology (2014)
- 8. Schulze F, Erdmann H, Hardkop LH, et al. Eruptive naevi and darkening of pre-existing naevi 24 h after a single mono-dose injection of melanotan II. European Journal of Dermatology (2014)
- 9. Mallory CW, Lopategui DM, Cordon BH. Melanotan tanning injection: a rare cause of priapism. Sexual Medicine (2021)
- 10. Habbema L, Halk AB, Neumann M, Bergman W. Risks of unregulated use of alpha-melanocyte-stimulating hormone analogues. International Journal of Dermatology (2017)
- 11. Minakova E, Lang J, Medel-Matus JS, et al. Melanotan-II reverses autistic features in a maternal immune activation mouse model of autism. Translational Psychiatry (2019)
- 12. Hadley ME, Dorr RT. Melanocortin peptide therapeutics: historical milestones, clinical studies and commercialization. Peptides (2006)
- 13. Wikipedia contributors. Melanotan II. Wikipedia, The Free Encyclopedia (2026)
Related Dosing Protocols
Educational use only
This guide is an educational research reference, not medical advice or a treatment plan. Melanotan 2 is not FDA-approved and is illegal to sell for human use in the US, UK, and Australia.
Calculate vial math
Use the PepPal calculator for custom vial size, BAC water volume, and syringe-unit math.
Open CalculatorWritten by Garret Grant
Founder & Lead Researcher · B.S. Civil Engineering, UCLA
Last updated: May 2026
Human-researched and AI-assisted with full editorial review. I verify sources, protocol interpretation, and final judgments personally. See methodology.
Share this page

