Pinealon Quick Start
Pinealon is a synthetic three-amino-acid peptide. Its sequence is Glu-Asp-Arg, which is why it is also called the EDR peptide. It is one of the short peptide bioregulators developed by Vladimir Khavinson's group at the St. Petersburg Institute of Bioregulation and Gerontology in Russia.
In the research community, Pinealon is mainly studied for brain aging, memory, and nerve-cell stress. The goal is to see whether it can help brain cells handle oxidative stress and support learning or focus during a short research cycle. These claimed benefits are still experimental and have not been proven in large human trials.
This page covers both oral and subcutaneous research planning, the vial math for the common 20 mg vial, and the current safety and regulatory standing. It is not a treatment plan and is not medical advice.
Route
Most research planning uses oral capsules or subcutaneous injection from a reconstituted vial.
Common vial
20 mg lyophilized powder. Reconstitute with bacteriostatic water.
Math anchor
20 mg vial + 3.0 mL BAC water = about 6.67 mg/mL. On a U-100 syringe, 1 unit equals 0.01 mL, or about 66.7 mcg.
Cycle length
Research cycles in the literature run roughly 10 to 20 days, repeated 2 to 3 times per year.
Status
Not FDA-approved for any human use as of May 2026.
Educational reference
This page is a research-context summary, not personal medical advice. Pinealon is sold under research-use-only labeling and is not an FDA-approved treatment. Talk to a qualified clinician before any peptide use.
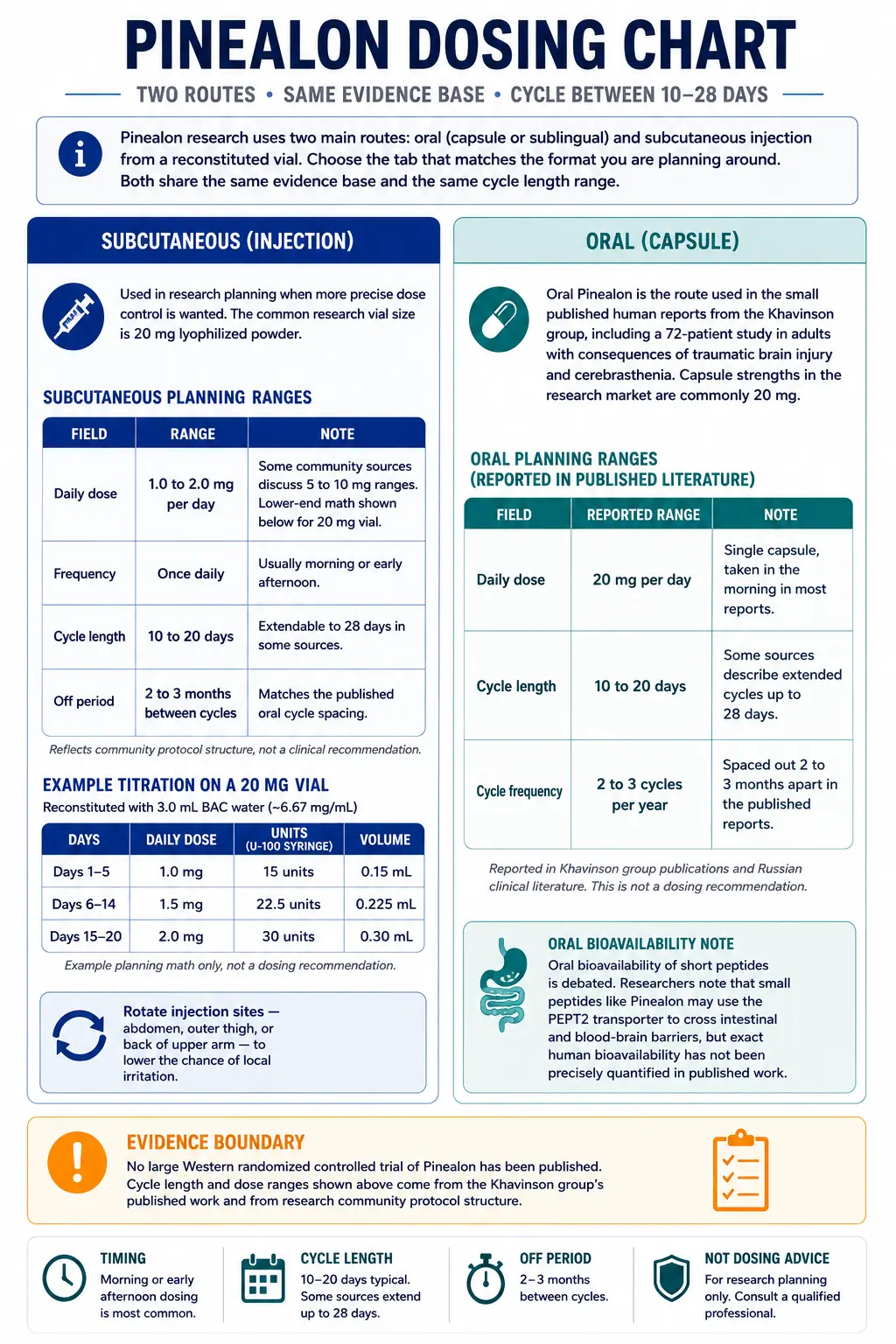
Pinealon Dosing Protocol & Schedule
Pinealon research uses two main routes: oral (capsule or sublingual) and subcutaneous injection from a reconstituted vial. Choose the tab that matches the format you are planning around. Both share the same evidence base and the same cycle length range.
Pinealon Protocol Formats
Choose the format you are researching to see route-specific notes.
Used in research planning when more precise dose control is wanted.
Subcutaneous injection is the more common route in research-use-only vial-based planning. The common research vial size is 20 mg lyophilized powder. Reconstitution math below is built around this vial.
Subcutaneous planning ranges discussed in research community protocols
Field
Daily dose
Range
1.0 to 2.0 mg per day
Note
Some community sources discuss 5 to 10 mg ranges. Lower-end math is shown below for the 20 mg vial.
Field
Frequency
Range
Once daily
Note
Usually morning or early afternoon in research community protocols.
Field
Cycle length
Range
10 to 20 days
Note
Extendable to 28 days in some sources.
Field
Off period
Range
2 to 3 months between cycles
Note
Matches the published oral cycle spacing.
| Field | Range | Note |
|---|---|---|
| Daily dose | 1.0 to 2.0 mg per day | Some community sources discuss 5 to 10 mg ranges. Lower-end math is shown below for the 20 mg vial. |
| Frequency | Once daily | Usually morning or early afternoon in research community protocols. |
| Cycle length | 10 to 20 days | Extendable to 28 days in some sources. |
| Off period | 2 to 3 months between cycles | Matches the published oral cycle spacing. |
Reflects community protocol structure, not a clinical recommendation.
Example titration on a 20 mg vial reconstituted with 3.0 mL BAC water (~6.67 mg/mL)
Example planning titration only
Days
Days 1–5
Daily dose
1.0 mg
Units on U-100 syringe
15 units
Volume
0.15 mL
Days
Days 6–14
Daily dose
1.5 mg
Units on U-100 syringe
22.5 units
Volume
0.225 mL
Days
Days 15–20
Daily dose
2.0 mg
Units on U-100 syringe
30 units
Volume
0.30 mL
| Days | Daily dose | Units on U-100 syringe | Volume |
|---|---|---|---|
| Days 1–5 | 1.0 mg | 15 units | 0.15 mL |
| Days 6–14 | 1.5 mg | 22.5 units | 0.225 mL |
| Days 15–20 | 2.0 mg | 30 units | 0.30 mL |
Example planning math only, not a dosing recommendation.
Rotate injection sites — abdomen, outer thigh, or back of upper arm — to lower the chance of local irritation.
The route used in most published Russian-language clinical reports.
Oral Pinealon is the route used in the small published human reports from the Khavinson group, including a 72-patient study in adults with consequences of traumatic brain injury and cerebrasthenia. Capsule strengths in the research market are commonly 20 mg.
Oral planning ranges reported in the published literature
Field
Daily dose
Reported range
20 mg per day
Note
Single capsule, taken in the morning in most reports.
Field
Cycle length
Reported range
10 to 20 days
Note
Some sources describe extended cycles up to 28 days.
Field
Cycle frequency
Reported range
2 to 3 cycles per year
Note
Spaced out 2 to 3 months apart in the published reports.
| Field | Reported range | Note |
|---|---|---|
| Daily dose | 20 mg per day | Single capsule, taken in the morning in most reports. |
| Cycle length | 10 to 20 days | Some sources describe extended cycles up to 28 days. |
| Cycle frequency | 2 to 3 cycles per year | Spaced out 2 to 3 months apart in the published reports. |
Reported in Khavinson group publications and Russian clinical literature. This is not a dosing recommendation.
Oral bioavailability of short peptides is debated. Researchers note that small peptides like Pinealon may use the PEPT2 transporter to cross intestinal and blood-brain barriers, but exact human bioavailability has not been precisely quantified in published work.
Evidence boundary
No large Western randomized controlled trial of Pinealon has been published. Cycle length and dose ranges shown above come from the Khavinson group's published work and from research community protocol structure.
Pinealon Supplies Needed
Math below assumes the common 20 mg subcutaneous vial reconstituted with 3.0 mL BAC water and once-daily dosing.
Recommended Supply
Use discount code PEPPAL at eligible peptide supplier checkouts.

Pinealon 20 mg Vials

BAC Water
Injection Supplies
Disclosure: supply links may earn PDP a commission at no cost to you.
Peptide Vials (20 mg each)
Each 20 mg vial covers roughly 10 days at the upper end of the example titration above.
| Cycle length | Planning note |
|---|---|
10 days 1 vial | About 10 daily draws at the example titration. |
20 days 2 vials | Roughly 20 daily draws. |
28 days 3 vials | Adds margin for priming losses or a longer cycle. |
10 days
1 vial
About 10 daily draws at the example titration.
20 days
2 vials
Roughly 20 daily draws.
28 days
3 vials
Adds margin for priming losses or a longer cycle.
Insulin Syringes (U-100, 0.3 or 0.5 mL)
One fresh syringe per injection. The example doses above fit a 0.3 mL or 0.5 mL barrel.
| Cycle length | Planning note |
|---|---|
10 days 10 syringes | 1 syringe per day. |
20 days 20 syringes | 1 syringe per day. |
28 days 28 syringes | 1 syringe per day. |
10 days
10 syringes
1 syringe per day.
20 days
20 syringes
1 syringe per day.
28 days
28 syringes
1 syringe per day.
Bacteriostatic Water
Use 3.0 mL per 20 mg vial. A standard 10 mL bottle covers up to 3 vials with margin.
| Cycle length | Planning note |
|---|---|
10-28 days 1 × 10 mL bottle | 10 days: Uses 3 mL of one bottle.; 20 days: Uses 6 mL of one bottle.; 28 days: Uses 9 mL of one bottle; close to the limit, so order a spare. |
10-28 days
1 × 10 mL bottle
10 days: Uses 3 mL of one bottle.; 20 days: Uses 6 mL of one bottle.; 28 days: Uses 9 mL of one bottle; close to the limit, so order a spare.
Numbers round up for priming losses, dropped syringes, and small protocol changes. Confirm vial size and current pricing on the supplier page before ordering.
Pinealon Reconstitution Guide
Reconstitution is the step where dry, lyophilized Pinealon powder is mixed with bacteriostatic water (BAC water) so it can be drawn into a syringe. For the common 20 mg vial, the example math below uses 3.0 mL of BAC water. This works out to about 6.67 mg/mL of Pinealon in solution.
Reconstitution math for the 20 mg Pinealon vial
BAC water added
2.0 mL
Concentration
10.0 mg/mL
1 unit (U-100) equals
0.10 mg (100 mcg)
BAC water added
3.0 mL
Concentration
6.67 mg/mL
1 unit (U-100) equals
0.067 mg (66.7 mcg)
BAC water added
4.0 mL
Concentration
5.0 mg/mL
1 unit (U-100) equals
0.05 mg (50 mcg)
| BAC water added | Concentration | 1 unit (U-100) equals |
|---|---|---|
| 2.0 mL | 10.0 mg/mL | 0.10 mg (100 mcg) |
| 3.0 mL | 6.67 mg/mL | 0.067 mg (66.7 mcg) |
| 4.0 mL | 5.0 mg/mL | 0.05 mg (50 mcg) |
Pick a volume that keeps your planned dose on an easy-to-read unit mark.
- 01
Wipe the vial top
Use a fresh alcohol swab on the rubber stopper of both the Pinealon vial and the BAC water vial.
- 02
Draw BAC water
Use a clean syringe to draw 3.0 mL of bacteriostatic water from its vial.
- 03
Add slowly
Insert the needle into the Pinealon vial and let the water run down the inside wall of the glass, not directly onto the powder.
- 04
Swirl, do not shake
Gently swirl or roll the vial between your hands until the powder dissolves into a clear solution. Skipping this can damage the peptide.
- 05
Inspect
The reconstituted solution should be clear and free of cloudy particles. If it looks off, do not use the vial.
- 06
Label and refrigerate
Write the reconstitution date on the vial. Store at 2 to 8 °C (35.6 to 46.4 °F).
- 07
Draw your dose
Use a fresh U-100 insulin syringe for each session. Match the unit mark to the table above for your chosen concentration.
Need custom vial math?
If your vial size, BAC water volume, or target dose is different, use the calculator for exact unit conversions.
Pinealon Dosage Chart
This Pinealon dosage chart summarizes the example subcutaneous titration schedule from 1 mg up to 2 mg, with dose escalation shown by day range.
How Pinealon Works
Pinealon is unusually small. At about 418 g/mol it is one of the smallest peptides used in neurological research. Khavinson's group has proposed that this small size lets Pinealon pass through cell membranes, including the nuclear membrane, and interact directly with DNA.
In plain English: most peptides plug into a receptor on the cell surface and trigger a signal from there. Pinealon is proposed to act more like a small key that slips inside the cell, reaches the nucleus, and helps switch certain genes on or off. The genes involved are linked to antioxidant defense and neuron survival.
The technical name for this proposed effect is epigenetic regulation through gene promoter binding. The leading hypothesis is that the Glu-Asp-Arg sequence binds to promoter regions and helps the cell make more of certain proteins. This mechanism is supported by preclinical work but has not been confirmed in large human trials.
Reduces reactive oxygen species
In rat cerebellar granule cells, neutrophils, and PC12 cells, Pinealon limited the buildup of reactive oxygen species under oxidative stress (Khavinson et al., Rejuvenation Research, 2011).
Modulates ERK 1/2 signaling
The same study described a delayed activation of the ERK 1/2 signaling pathway and changes in the cell cycle.
Restores dendritic spines
In a mouse hippocampal neuron model of Alzheimer's amyloid toxicity, EDR (200 ng/mL) increased mushroom dendritic spines by 71%, returning the parameter close to normal (Kraskovskaya et al., 2017).
Who Pinealon Is For and Who Should Avoid It
Pinealon is not approved for treatment. Even in research planning, some groups should not be exposed to it because the safety data is too thin or the theoretical risk is too high.
- Pregnancy and breastfeeding: safety has not been established. Avoid.
- Active cancer or recent cancer history: Pinealon affects apoptosis and caspase-3 signaling. Because caspase-3 has been linked to both tumor suppression and, in some contexts, tumor support, this group should avoid Pinealon outside formal oncology research.
- Seizure disorders: any compound that acts on the central nervous system should be approached cautiously here.
- Children and teenagers: no safety data exists in this group.
- Anyone on prescription central nervous system medications: drug interactions are not formally mapped.
- Known peptide allergies: prior reactions to any short peptide are a reason to skip it.
If any of those apply, talk to a qualified clinician before considering Pinealon for any reason.
Pinealon Side Effects & Safety
Published Pinealon research describes the peptide as generally well tolerated. The side-effect record is thinner than for FDA-approved drugs, so the picture below should be read as what has been reported, not what is known to be safe long-term.
Most commonly reported
- Mild headache, especially early in a cycle.
- Sleep changes (vivid dreams, lighter or deeper sleep) during the first week.
- Mild gastrointestinal effects with oral capsules.
- Injection-site redness or itching with subcutaneous use.
Theoretical or under-studied
- Allergic reaction (any peptide can cause one).
- Long-term effects of repeated cycles have not been mapped in published Western trials.
- Pinealon's effect on caspase-3 signaling means cancer-related risk has not been ruled out in or out — researchers in oncology contexts generally exclude it.
Quality-control risks
Because Pinealon is sold under research-use-only labels, batch purity, sequence identity, and contamination depend on the supplier. Match the certificate of analysis (COA) to the exact vial and lot before any use. Treat poorly documented vials as the bigger near-term risk, not the peptide itself.
Pinealon Timeline & What to Monitor
Subjective changes reported in research cycles tend to appear after 10 to 14 days of daily use. The 72-patient TBI cohort study reported memory and headache improvements during a course of oral treatment combined with standard therapy, not as a single peak effect.
- Days 1–7: usually no clear subjective change. Sleep quality is the most common early observation.
- Days 10–14: most reported subjective changes (memory recall, daytime mental clarity, mood evenness) show up in this window.
- End of cycle (day 20–28): cycle ends. Off-period of 2 to 3 months between cycles in the published structure.
Pinealon has no validated lab marker for monitoring outside formal research. Most people running research-context protocols simply log subjective sleep, focus, and mood notes. Sudden new headaches, persistent gastrointestinal symptoms, or any allergic-type reaction are reasons to stop.
Pinealon Clinical Evidence Context
Most of the Pinealon evidence base is preclinical — cell culture and rat studies, mostly from Khavinson's group at the St. Petersburg Institute of Bioregulation and Gerontology. A small number of human reports exist in the Russian-language clinical literature, including a 72-patient study in adults with consequences of traumatic brain injury. No large randomized controlled trial of Pinealon has been published in the Western peer-reviewed literature.
Human (small, Russian-language)
Oral Pinealon plus standard therapy in 72 patients with consequences of traumatic brain injury and cerebrasthenia. Reported improvements in memory, headache duration, emotional balance, and performance.
Cell-culture
Dose-dependent reduction of reactive oxygen species and necrotic cell death in cerebellar granule cells, neutrophils, and PC12 cells (Khavinson et al., Rejuvenation Research, 2011).
Cell-culture (Alzheimer's model)
EDR at 200 ng/mL restored mushroom dendritic spines in mouse hippocampal neurons exposed to amyloid (Kraskovskaya et al., 2017).
Rat (prenatal hyperhomocysteinemia)
Pinealon improved offspring spatial learning and reduced oxidative damage in cerebellar neurons (Arutjunyan et al., 2012).
Rat (experimental diabetes)
Pinealon supported learning retention and altered hippocampal NMDA receptor subunit expression (Karantysh et al., Neurochemical Journal, 2020).
Key gap: no large Western randomized trial. Long-term safety data in humans is limited. Independent replication of the proposed direct-DNA-interaction mechanism outside Khavinson's group remains thin.
Pinealon Storage & Handling
Storage is one of the easiest places to lose peptide potency. Pinealon is sensitive to heat, light, and repeated freeze-thaw cycles.
Pinealon storage temperatures
Form
Lyophilized powder (sealed vial)
Temperature
-4 °F (-20 °C)
Note
Best for long-term storage. Keep dry and dark.
Form
Lyophilized powder (short-term)
Temperature
35.6 to 46.4 °F (2 to 8 °C)
Note
Acceptable for short transit windows.
Form
Reconstituted (in BAC water)
Temperature
35.6 to 46.4 °F (2 to 8 °C)
Note
Refrigerate. Use within the BAC water shelf window.
Form
Appearance
Temperature
Clear after mixing
Note
Cloudy or particulate solution means stop using it.
| Form | Temperature | Note |
|---|---|---|
| Lyophilized powder (sealed vial) | -4 °F (-20 °C) | Best for long-term storage. Keep dry and dark. |
| Lyophilized powder (short-term) | 35.6 to 46.4 °F (2 to 8 °C) | Acceptable for short transit windows. |
| Reconstituted (in BAC water) | 35.6 to 46.4 °F (2 to 8 °C) | Refrigerate. Use within the BAC water shelf window. |
| Appearance | Clear after mixing | Cloudy or particulate solution means stop using it. |
Avoid repeated freeze-thaw cycles. Let frozen vials reach room temperature before opening to reduce condensation.
Pinealon Protocol Mistakes & Troubleshooting
I missed a daily dose
Skip the missed dose and resume the next day. Do not double up.
The vial looks cloudy after mixing
Stop. Cloudiness can mean degradation, contamination, or undissolved powder. Reconstituted Pinealon should be clear.
I added the wrong amount of BAC water
Recalculate the concentration. The vial is now at a different mg/mL, so your unit math also changes. Use the calculator before the next draw.
I forgot to refrigerate the reconstituted vial
If it sat at room temperature for more than a few hours, treat the vial as compromised.
Mild headache started on day two
Reported in some research community logs as an early-cycle effect that fades. Stop if it intensifies or persists.
I am mixing routes mid-cycle
Pick oral or subcutaneous and stay with it for the whole cycle. Switching mid-cycle makes the dose math confusing and obscures any subjective changes.
Pinealon Regulatory Status
As of May 2026, Pinealon is not FDA-approved for any human indication in the United States. It is not on the FDA's Category 1 (permissible for compounding) bulk drug substance list under Sections 503A or 503B of the Federal Food, Drug, and Cosmetic Act. It is not available through licensed U.S. compounding pharmacies by prescription.
Products labeled as Pinealon and sold by online vendors are usually marketed as research use only. They are not regulated as drugs or as dietary supplements. Quality, identity, and purity depend on the supplier.
In Russia, Pinealon has a longer regulatory history through a separate framework for short peptide bioregulators. That status does not carry over to the United States.
Status checked May 2026
Regulatory status can change. Confirm current FDA Category 1 listing and 503A/503B nomination status on FDA.gov before relying on this paragraph for planning.
Pinealon vs Epitalon vs Semax vs Selank
Pinealon sits in the same family as Khavinson's other short peptide bioregulators and is often compared to neuropeptides from a different Russian research line.
Short brain-active peptide comparison
Peptide
Pinealon
Sequence
Glu-Asp-Arg (3 aa)
Primary research focus
Neuroprotection, cognition, oxidative-stress endpoints
Common route
Oral or subcutaneous
Peptide
Epitalon
Sequence
Ala-Glu-Asp-Gly (4 aa)
Primary research focus
Pineal gland, telomerase, longevity endpoints
Common route
Subcutaneous
Peptide
Semax
Sequence
ACTH(4-7) analog (7 aa)
Primary research focus
Cognition, focus, neuroprotection
Common route
Intranasal
Peptide
Selank
Sequence
Tuftsin analog (7 aa)
Primary research focus
Anxiety, cognition, immune modulation
Common route
Intranasal
| Peptide | Sequence | Primary research focus | Common route |
|---|---|---|---|
| Pinealon | Glu-Asp-Arg (3 aa) | Neuroprotection, cognition, oxidative-stress endpoints | Oral or subcutaneous |
| Epitalon | Ala-Glu-Asp-Gly (4 aa) | Pineal gland, telomerase, longevity endpoints | Subcutaneous |
| Semax | ACTH(4-7) analog (7 aa) | Cognition, focus, neuroprotection | Intranasal |
| Selank | Tuftsin analog (7 aa) | Anxiety, cognition, immune modulation | Intranasal |
These are not interchangeable. Each has its own evidence base and its own safety boundary.
Pinealon is closest to Epitalon in research lineage (both come from Khavinson's group, both are short bioregulators) but differs in target tissue and sequence. Semax and Selank are from a separate Russian research line, use a different route (intranasal), and target different endpoints.
FAQ
Q1: What is Pinealon?
Pinealon is a synthetic three-amino-acid peptide with the sequence Glu-Asp-Arg, also called the EDR peptide. It was developed by Vladimir Khavinson's research group at the St. Petersburg Institute of Bioregulation and Gerontology in Russia and is studied for neuroprotection, cognition, and cellular stress endpoints.
Q2: Is Pinealon FDA-approved?
No. As of May 2026, Pinealon has no FDA-approved indication and is not on the FDA Category 1 (permissible for compounding) bulk drug substance list under sections 503A or 503B. It is sold under research-use-only labeling outside Russia.
Q3: How is Pinealon dosed in research planning?
Published Russian-language reports used 20 mg per day orally for 10–20 days. Research community protocols for subcutaneous use commonly start around 1.0–2.0 mg per day from a reconstituted 20 mg vial. This is research-context structure, not a dosing recommendation.
Q4: Can Pinealon be taken orally?
Yes — oral capsules at around 20 mg per day are the route used in most published human reports from the Khavinson group. Researchers have proposed that the small tripeptide may use the PEPT2 transporter to cross intestinal and blood-brain barriers, but exact human bioavailability has not been precisely quantified.
Q5: How is Pinealon reconstituted?
A common research approach is to add 3.0 mL of bacteriostatic water to a 20 mg lyophilized vial, giving roughly 6.67 mg/mL. On a U-100 insulin syringe, 1 unit then equals about 66.7 mcg of Pinealon. Always swirl, do not shake, and refrigerate after mixing.
Q6: How long is a typical Pinealon cycle?
Published cycles run 10 to 20 days, sometimes extended to 28 days, and are usually repeated 2 to 3 times per year. The off-period between cycles is typically 2 to 3 months in the Khavinson group's reports.
Q7: What supplies are needed for a Pinealon cycle?
For a subcutaneous 10–20 day cycle from a 20 mg vial: 1–2 vials, one fresh U-100 insulin syringe per day, one 10 mL bottle of bacteriostatic water, and a 100-count box of alcohol swabs. See the supplies section above for exact math by cycle length.
Q8: What are the main Pinealon side effects?
Most published research describes Pinealon as well tolerated. Reported effects include mild headache early in a cycle, sleep changes, mild gastrointestinal effects with oral use, and injection-site redness with subcutaneous use. Long-term safety data in humans is limited.
Q9: Who should avoid Pinealon?
Pregnant or breastfeeding people, anyone with active cancer or a recent cancer history, anyone with a seizure disorder, children and teenagers, and anyone on prescription central nervous system medications should avoid Pinealon outside formal medical supervision.
Q10: How does Pinealon compare to Epitalon?
Both come from Khavinson's research group, but Pinealon is a tripeptide (Glu-Asp-Arg) focused on neuroprotection and cognition, while Epitalon is a tetrapeptide (Ala-Glu-Asp-Gly) focused on pineal gland, telomerase, and longevity endpoints. They are not interchangeable.
Q11: Has Pinealon been studied in humans?
Yes, but in small, mostly Russian-language reports. The best-known is a 72-patient study using oral Pinealon alongside standard therapy in adults with consequences of traumatic brain injury. No large Western randomized controlled trial of Pinealon has been published.
Q12: Is this page medical advice?
No. This page is an educational research-context reference. Pinealon is not FDA-approved for any human use. Talk to a qualified clinician before considering any peptide.
Sources & Research
- 1. Khavinson V, Ribakova Y, Kulebiakin K, et al. Pinealon increases cell viability by suppression of free radical levels and activating proliferative processes. Rejuvenation Research (2011)
- 2. Kraskovskaya NA, Kukanova EO, Lin'kova NS, Popugaeva EA, Khavinson VKh. Tripeptides Restore the Number of Neuronal Spines under Conditions of In Vitro Modeled Alzheimer's Disease. Bulletin of Experimental Biology and Medicine (2017)
- 3. Khavinson VKh, Lin'kova NS, Kvetnoy IM, et al. EDR Peptide: Possible Mechanism of Gene Expression and Protein Synthesis Regulation Involved in the Pathogenesis of Alzheimer's Disease. Molecules (MDPI) (2021)
- 4. Arutjunyan A, Kozina L, Stvolinskiy S, et al. Pinealon protects the rat offspring from prenatal hyperhomocysteinemia. International Journal of Clinical and Experimental Medicine (2012)
- 5. Karantysh GV, Mendzheritskii AM, Ryzhak GA, et al. Effect of Pinealon on Learning and Expression of NMDA Receptor Subunit Genes in the Hippocampus of Rats with Experimental Diabetes. Neurochemical Journal (2020)
- 6. Khavinson VKh, Tarnovskaya SI, Lin'kova NS, et al. Short Peptides and Telomere Length Regulator Hormone Irisin. Bulletin of Experimental Biology and Medicine (2016)
- 7. Khavinson VKh, Lin'kova NS, Umnov RS. Peptide KED: Molecular-Genetic Aspects of Neurogenesis Regulation in Alzheimer's Disease. Bulletin of Experimental Biology and Medicine (2021)
- 8. Meshchaninov VN, Tkachenko EL, Zharkov SV, et al. Effect of synthetic peptides on aging of patients with chronic polymorbidity and organic brain syndrome of the central nervous system in remission. Advances in Gerontology (2015)
- 9. National Center for Biotechnology Information. PubChem Compound Summary for CID 10273502, Pinealon (Glu-Asp-Arg). PubChem (2026)
- 10. U.S. Food and Drug Administration. Bulk Drug Substances Nominated for Use in Compounding Under Section 503A (Category 1 list). FDA.gov (2026)
Related Dosing Protocols
Educational use only
This guide is an educational research reference, not medical advice or a treatment plan. Pinealon is not FDA-approved for human use.
Calculate vial math
Use the calculator for custom vial size, BAC-water volume, and syringe-unit math.
Open CalculatorWritten by Garret Grant
Founder & Lead Researcher · B.S. Civil Engineering, UCLA
Last updated: May 2026
Human-researched and AI-assisted with full editorial review. I verify sources, protocol interpretation, and final judgments personally. See methodology.
Share this page
